Surface Plasmon Resonance
Pulsatile-free flow
Stable and steady flow profile
Vibration-free pump
Ideal for sensitive experiments near the microscope
Compact and modular
Small footprint and several channels in parallel

Need a microfluidic SME partner for your Horizon Europe project?
Surface plasmon resonance
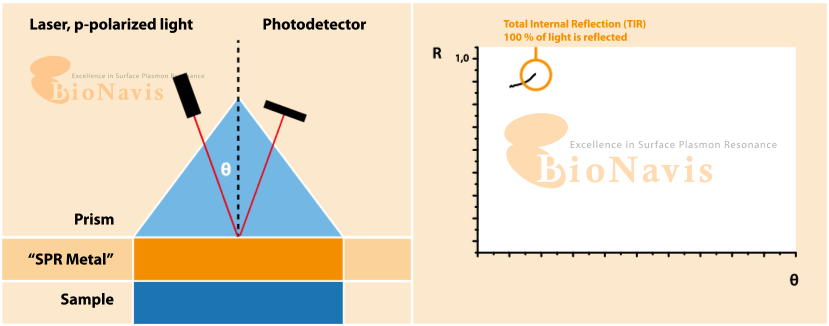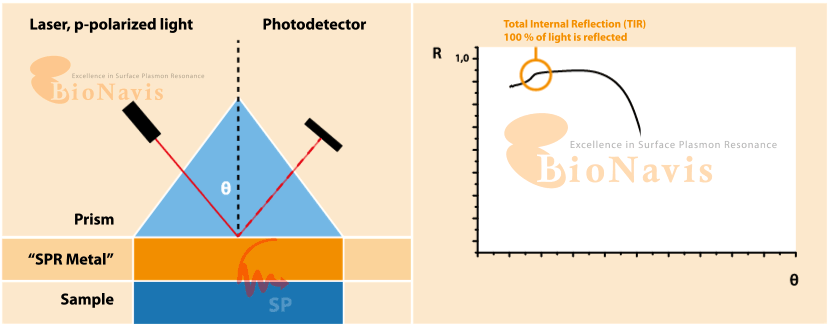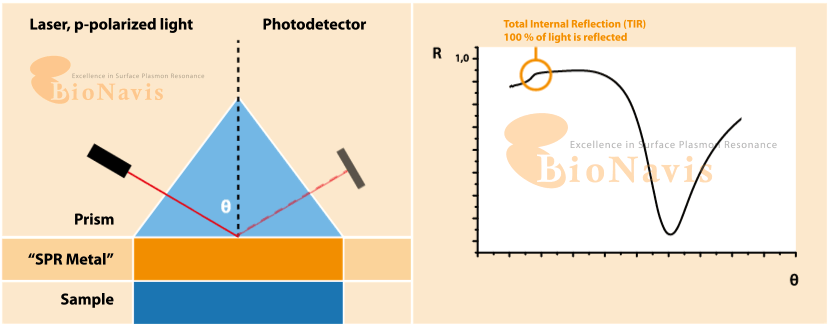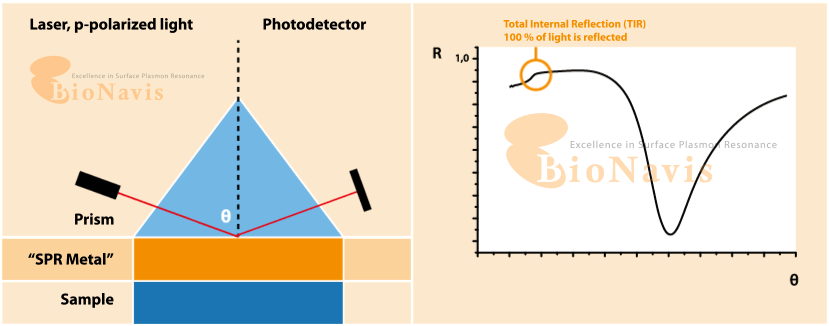
Surface plasmon resonance (SPF) is an optical technique for sensing alterations on a treated surface. The working principle of SPF consists in applying a light beam at an angle on a metallic surface and reading the difference between the expected refractive angle and the actual refractive angle, which characterises the surface plasmon resonance. The technique is highly sensitive, and can detect small changes on the surface, such as the binding of molecules.
Our Perfusion pump was designed precisely to address these limitations. Compact and easy to assemble and use, it offers a stable and steady flow, at low flow rates, vibration-free. The working volumes can be easily adapted from 1,5 ml Eppendorfs to 100ml bottles.
Surface plasmon resonance-based cell sensing
SPR can be used for cell sensing. For example, it is possible to detect how cells attach, molecule-ligand studies, with either attached cells or in suspension.
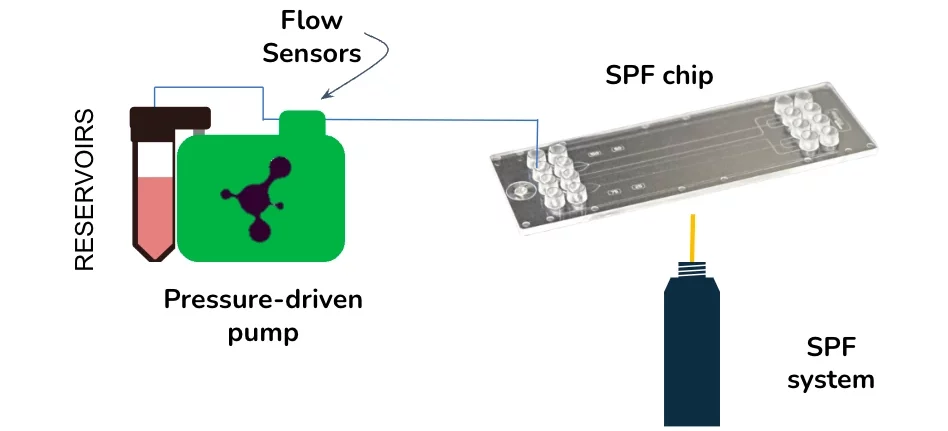
Setup
The surface plasmon resonance setup remains the same with the perfusion pump replacing the syringe pumps. Although the perfusion pump is pressure-based, requiring a flow sensor to control low flow rates, the flow sensor and reservoirs are integrated, making it easier to use.
References
Annika Koponen, Erja Kerkelä, Tatu Rojalin, Elisa Lázaro-Ibáñez, Teemu Suutari, Heikki O. Saari, Pia Siljander, Marjo Yliperttula, Saara Laitinen, Tapani Viitala, Label-free characterization and real-time monitoring of cell uptake of extracellular vesicles, Biosensors and Bioelectronics, Volume 168, 2020, 112510, ISSN 0956-5663, https://doi.org/10.1016/j.bios.2020.112510.
Compatibility and applications
Some biological applications of our perfusion pump for microscopy also include:
Biomolecular Interaction Analysis
Drug Discovery and Development
Biosensor Diagnostic Development
Cell attachment studies
And many more!

References
1. Lyapun, I. N., Andryukov, B. G. & Bynina, M. P. HeLa Cell Culture: Immortal Heritage of Henrietta Lacks. Mol. Genet. Microbiol. Virol. 34, 195–200 (2019).
2. Coluccio, M. L. et al. Microfluidic platforms for cell cultures and investigations. Microelectron. Eng. 208, 14–28 (2019).
3. Mehling, M. & Tay, S. Microfluidic cell culture. Curr. Opin. Biotechnol. 25, 95–102 (2014).
Perfusion pump technical specifications
| Pressure control | |
|---|---|
| Pressure range | -400 to 600 mbar |
| Pressure stability | 0.2 mbar |
| Air flow rate | 0.1 L/min at atmospheric pressure
Possibility to work with higher air flow rates by reducing the pressure range |
| Flow control | |
| Microfluidic flow sensor | Monitoring and feedback loop flow control available |
| Flow rates | From 0.1 µL/min to 5 mL/min |

Frequently asked questions
Which microscope slides can be used in Surface Plasmon Resonance?
Our system is designed to adapt to any chips or chambers, depending only on having the correct adapters.
Is the perfusion pump for microscopy powered by battery?
No, the perfusion pump for microscope needs to be connected to a power source.
Does the perfusion pump require an air compressed source?
No, the pump is autonomous.


Funding and Support
This project has received funding from the European Union’s Horizon research and innovation program under HORIZON-HLTH-2024-TOOL-05-two-stage, Grant agreement number 101155875 (NAP4DIVE).
Products & Associated Accessories
FAQ - Surface Plasmon Resonance
What is Surface Plasmon Resonance (SPR)?
SPR is a form of optical sensing technique that measures minute variations occurring at the surface of a metal (usually gold). When you apply light at a given angle, and when molecules bind, cells attach, or the local environment changes, the resonance condition shifts. Due to the extreme sensitivity of the readout, any disturbance in liquid flow (pulses, vibrations, micro-bubbles, etc.) is likely to be reflected in noise.
What are the major problems of the conventional SPR experimental systems?
Traditional SPR experiments are usually performed using syringe pumps to dispense liquid over the metallic sensing surface. Although syringe pumps are recommended to contain low flow rates and are also simple to operate, they have a number of limitations:
- Pulsatile flow profile: Noise is added to sensitive SPR readings due to mechanical motion that causes flow pulsations.
- Vibrations: Mechanical movements around the microscope generate vibrations, which necessitate dampening systems to reduce further noise.
- Bulky design: Syringe pumps take up a lot of bench space.
- Small working volume: The syringe’s size determines the volume, limiting the experiment’s flexibility.
- These limitations can compromise the quality and reliability of sensitive SPR measurements.
What are the technical specification of the Perfusion Pump?
Pressure Control:
- Pressure range: -400 to 600 mbar
- Pressure stability: 0.2 mbar
- Air flow rate: 0.1 L/min and atmospheric pressure (it can be higher by decreasing pressure range)
Flow Control:
- Microfluidic flow sensor: Visualizing and control feedback loop.
- Flow rates: 0.1 uL/min to 5 mL/min
- Control mechanism: Pressure-based having inbuilt flow sensors.
Power Requirements:
- Needs to be connected to power source (can not be battery-powered).
- Independent working (no source of compressed air is needed)
What is Surface Plasmon Resonance as a Cell Sensor?
Functions SP can be used to apply to cell sensing.
- Cell attachment experiments: Sensing cell adhesion to bioreceptor surfaces.
- Molecule-ligand interactions: Investigating the binding phenomenon with either cell attached or suspended cells.
- Live cell observation: Dynamics and cellular interactions in real time.
- Label-free characterization: Watershed-free tracking of cell uptake of extracellular vesicles without utilising fluorescent or radioactive labels.
That is why SPR can be highly useful when examining cellular behavior and interactions under physiologically significant conditions.
How does a normal experimental layout of SPR work with the Perfusion Pump?
The system incorporates the perfusion pump in regular SPR systems:
Components:
- Gold-coated slide (usually metallic) sensing surface.
- Perfusion pump used in lieu of traditional syringe pumps.
- Flow sensors and reservoirs all in one.
- Refractometric angle optical detector.
- Microfluidic flow chamber or microfluidic chip.
Benefits:
The pressure-based control of the perfusion pump that has inbuilt flow sensors ensures that the ease of use is retained without the problem of pulsation and vibration as was experienced with mechanical syringe pumps.
To what extent is it microscope-friendly (noise, vibration, footprint)?
The pack is tailored for delicate experiments near the microscope: non-vibrational, with a non-pulsatile flow profile, modular, and compact. You will know why this is important if you ever have to design a dampening system to ensure that the syringe pump’s vibrations do not reach your optical display.
What are the maximum reservoir volumes that you can operate, and is this a limitation in experiment design?
Working volume is flexible. The system is scalable to a small 1.5 mL microtube (Eppendorf-style) and to larger volumes, e.g., 100 mL bottles. That is a useful range of short kinetic runs using valuable reagents, up to longer experiments with increased buffer stability without frequent refills.
What chips, slides, or flow chambers can be used with the SPR pack?
The system is meant to be universal: compatibility is driven by the presence of the right connectors/adapters for your chip or chamber. It is not locked up to a single proprietary format; however, provided your fluidic interfaces can be cleanly adapted, the pack can often be integrated.
What are some common uses of this setup besides classical biomolecular binding?
The apparent ones are biomolecular interaction analysis, drug discovery and development, and biosensor development. However, SPR-based cell sensing is also applicable to the pack: cell attachment, ligand studies on adherent cells, or a cell-in-suspension assay. Anything that requires quietness and steady perfusion across the sensing area is likely to be useful.
Does it require an external compressed air source, and is it battery-operated?
No remote compressed-air source is needed; the pump is autonomous. Conversely, it does not run on a battery. That trade-off is quite common when you are more concerned with stability and control than with portability.

