CO₂ Control for Dynamic Cell Culture
CO₂ sensing and control independent of a CO₂ incubator
Be independent of the CO₂ Incubator
Control CO₂ of the media on the bench
Use the chip of your preference
Inline sensor can be added to any setup
Automated cell culture
Less manual work and more accuracy for your experiments
Plug-and-play platform
Beginner friendly pack with detailed user guide

Need a microfluidic SME partner for your Horizon Europe project?
The importance of CO₂ control for cell culture
The human body maintains its pH through a buffering system based on bicarbonate and CO2. In other words, CO2, usually produced as waste from metabolism, reacts with H2O to form carbonic acid, a weak acid. This reaction creates a dynamic equilibrium that shifts depending on the concentration of H+ to maintain the overall concentration of ions constant, as shown below. The amount of CO2 and carbonate available to buffer the system is regulated by the lungs and kidneys according to the body’s needs.
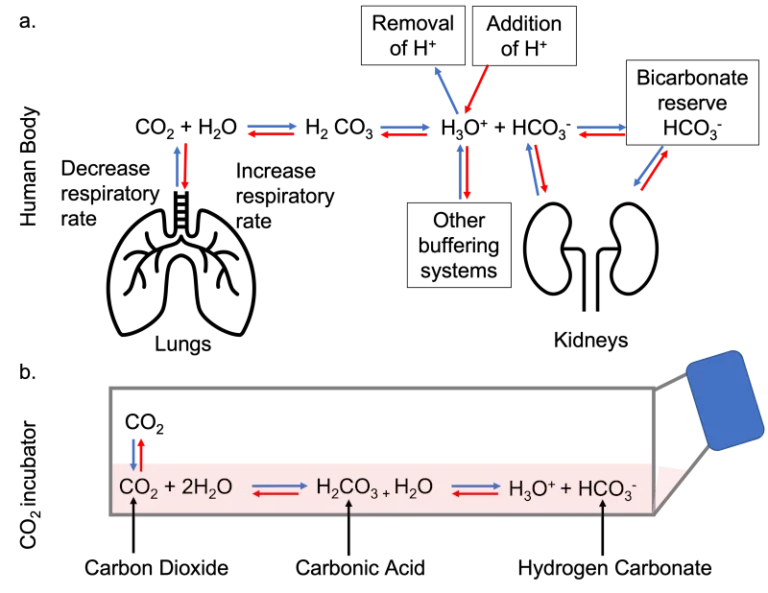
In cell culture, the same principle is applied. Media usually contains a predefined concentration of carbonate and the CO2 incubator maintains the internal atmosphere with a determined concentration of CO2, so the same dynamic equilibrium is formed.
However, there is no automatic way to adjust the concentrations of the buffering components as the body does with the lungs and the kidney, so the cell culture media needs to be changed regularly to keep the pH within adequate ranges.
The importance of CO₂ control in dynamic cell culture

In dynamic cell cultures, that is, when cells are cultured under flow, the microenvironment parameters become more important to control because the system’s small dimensions make small changes in pH more impactful.
Thus, either the entire perfusion system is designed to fit inside the CO2 incubator, or parts of the system (mainly the electronics) remain outside, and the chip with the cells is kept inside the incubator in a far-from-ideal configuration.
CO2 control in dynamic cell culture pack setup
Our all-in-one pack guarantees good compatibility between the different instruments, allows you to start your experiment right away, is piloted by a single software, and can be used for other applications. We also provide continuous and full customer support for you to fulfill your experiment goals.
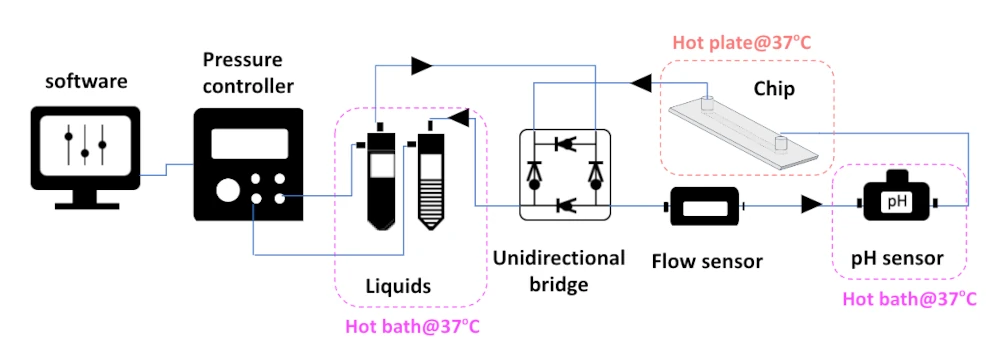
The pack contains:
Flow sensor (Galileo, MIC)
Software (Galileo user interface)
A pH/CO2 sensor and flow cell for use in line in setup
Pressure controller
Several Eppendorfs or Falcon reservoirs
Recirculation bridge
Microfluidic chips
Tubing and luers
User guide
This pack can also be combined with other microfluidic steps following the dynamic cell culture, such as sequential drug injection.
Behavior of CO2 in cell culture media
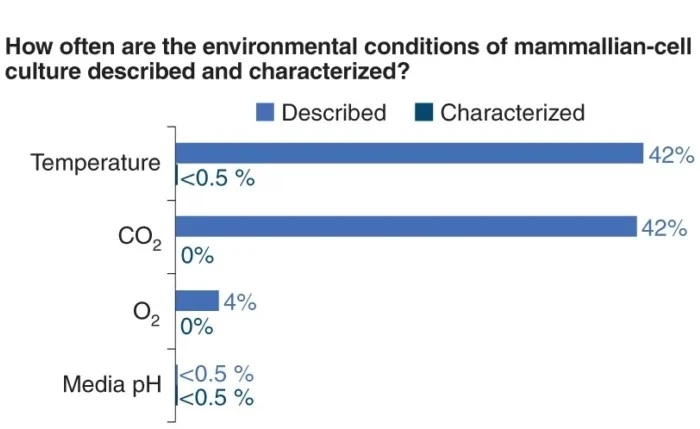
Culturing cells inside a CO2 incubator set at 5% has become such a standard practice in biology labs. A report from Klein et al. [1], analyzing 810 papers, clearly illustrates this. Most cell culture parameters, such as temperature, CO2 and O2 concentration and pH, are often described but rarely characterized or measured during experiments.
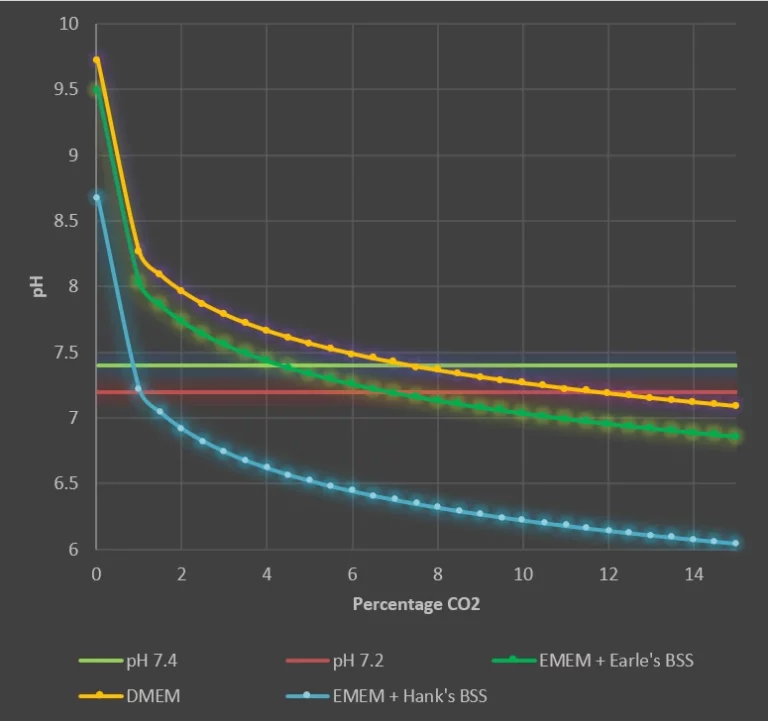
Consequently, few realize that different cell culture media require different CO2 levels to maintain the pH within acceptable ranges, as illustrated below. DMEM, for example, has 44 mM of NaHCO3, requiring a higher concentration of CO2 to achieve physiological pH [2].
References
Klein, S.G., Alsolami, S.M., Steckbauer, A. et al. A prevalent neglect of environmental control in mammalian cell culture calls for best practices. Nat Biomed Eng 5, 787–792 (2021). https://doi.org/10.1038/s41551-021-00775-0
Michl, J., Park, K. C. & Swietach, P. Evidence-based guidelines for controlling pH in mammalian live-cell culture systems. Commun. Biol. 2019 21 2, 1–12 (2019).
Cooper, J. CO2 concentration and pH control in the cell culture laboratory. Culture Collections, 2019. Accessed on 22nd November 2022.
Lu, C. & Verbridge, S. S. Microfluidic methods for molecular biology. Microfluid. Methods Mol. Biol. 1–376 (2016). doi:10.1007/978-3-319-30019-1
Customize your pack
This pack is highly customizable to fit your experiment specification and cell lines (mammalian, plant, primary…).
You can request additional pumping channels for the flow controller and flow rate sensors. Bubbles can be a problem for cells; the pack can contain a bubble remover to tackle this issue efficiently.
You can contact our experts to answer any questions about this pH control in the cell culture pack and how it can match your specifications.
Frequently asked questions
How can we help your experiment?
This pack is in beta testing phase. So, although the instruments are not fully industrialized, we can provide extensive support as part of our beta testing program. Get in touch to see if you are eligible.
Can a pack be customized?
Yes! Our experts will establish which instruments are best suited for your application, such as the type of flow sensor or the number of flow controller channels you need to perform your experiment. Contact us using the “talk to our experts” green button above.
Can I buy individual instruments?
Our instruments are in beta testing phase and can be tested as a pack or individually, so get in contact with our team to know how our beta testing program works.





Funding and Support
The results of the Protomet, ALTERNATIVE, and LIFESAVER projects helped develop this instrument pack, with funding from the European Union’s Horizon 2020 MSCA-ITN under grant agreement No 813873 (ProtoMet), the European Union under H2020-LC-GD-2020-3, grant agreement No. 101036702 (ALTERNATIVE), and the European Union under H2020-LC-GD-2020-3, grant agreement No. 101037090 (LIFESAVER).
Products & Associated Accessories
FAQ - Microfluidics for CO₂ Control in Cell Culture
Why is CO2 regulation so important in cell culture?
In vivo, CO2 combines with water to produce carbonic acid, which is part of the bicarbonate buffering system that maintains physiological pH (7.2-7.4). This mechanism is emulated in cell culture media by a specified amount of carbonate in equilibrium with the CO2 atmosphere established by the incubator. CO2 drift, such as opening doors of an incubator, evaporation of media, or metabolic activity, may cause pH to fall or fall out of range, altering cell behavior, impairing data reproducibility, and, in difficult situations, resulting in cell death.
What is the most significant weakness of standard CO2 incubators over dynamic cell culture?
Traditional CO2 incubators are used in immobile cultures in well plates or flasks. When used in dynamic cell culture, in which cells are perfused in continuous flow, the microfluidic system extends much further than the incubator footprint. Electronics, flow controllers, and sensors are generally located outside, and this makes it a split arrangement, which is not ideal, to say the least. This compels researchers to make an inconvenient concession either by cramming electronics into the incubator or by accepting that the aspect of the system is in contact with ambient CO2 levels, which they are attempting to avoid.
What is different about the CO2 Control Pack?
The pack offers inline CO2 and pH measurements in the fluidic circuit, and the CO2 incubator can be operated independently. The system does not use the incubator atmosphere to establish equilibrium in the media, but instead measures the CO2 concentration and pH in real time as the fluid passes through the chip. This implies that the setup may be either located on a bench, under a microscope, or any other arrangement that is suitable for the experiment, without compromising environmental control.
What are the instruments and components in the pack?
The pack includes an all-in-one set that contains: a Galileo flow sensor and user interface software, a pressure-based flow controller, an inline pH/CO2 sensor and flow cell, various Eppendorf or Falcon reservoirs, a recirculation bridge, microfluidic chips, tubing and Luer connectors, and a user instruction manual. Whether the components are pre-assembled to match and can be controlled through a single software interface, making it possible for individuals with limited knowledge of microfluidics to have a plug-and-play, user-friendly setup.
Do all the culture media have the same CO2 level to maintain physiological pH?
No, and this is a very much neglected fact. Sodium bicarbonate concentrations in various media vary and change the required CO2 concentration to regulate the pH within the7.2-7.4 pH range. An example is that DMEM has 44 mM NaHCO3 and should be incubated in about 7.5-11 percent CO2 to maintain the physiological pH of the medium; thus, running DMEM in a standard 5 percent CO2 incubator would give an approximate pH of about 7.5, a bit higher than desired. Media prepared by Hank using his BSS, on the contrary, demand almost atmospheric CO2 levels. Trying to use inline sensing enables the researcher to set the right conditions for their particular medium, rather than the 5% standard.
What is the advantage of the recirculation feature to cell culture experiments?
The recirculation bridge permits culture media to be recirculated within the chip, rather than being flowed through in a single pass and wasted. This minimizes overall media exposure – critical with the use of both costly or limited supplements – as well as enables the stabilization of the chemical environment surrounding the cells during more extended culture times. It also maintains automated feedback processes in which CO2 or pH sensors indicate changes in flow rate or gas delivery, enhancing reproducibility without manual adjustment.
Is the pack compatible with any custom-designed microfluidic chip?
Yes. The inline sensor is incorporated into the fluidic circuit either above or below the chip, making it compatible with both commercially available chips and homemade versions. CO2 control can be used by researchers working on organ-on-chip models, perfusion chambers, or any other geometry without being limited to a particular chip geometry. Other microfluidic steps, such as sequential drug injection, can also be integrated into the pack, enabling the construction of more complex, multi-step culture platforms.
What are the cell types that this pack is supported on, and how configurable is the pack?
The pack is designed to adapt to mammalian, plant, and primary cell lines. Its customization features include the ability to add more flow controllers and sensor channels to conduct experiments with more than two inlets, a bubble-remover module to protect delicate cells and prevent exposure to air, and various reservoir options to accommodate different culture volumes. MIC specialists collaborate hands-on with users to set up the system for specific cell lines, media conditions, and time periods.
Is it possible to use MIC as a technical partner in a Horizon Europe consortium?
As a microfluidic SME, MIC is regularly involved in EU research consortia as a non-academic beneficiary/associated partner, providing hardware components, flow-control instrumentation, and measurement expertise. MIC can co-design work packages based on the prototype deliverables, give early demonstrators that build on the technical credibility of the proposal, and risk-proof manufacturable chip designs.
Consortia incorporating the prototype-first development model of MIC are more likely to have more realistic milestones and more obvious technical routes, which are associated with higher scores in the evaluation at the proposal stage. If you are creating a consortium around the microfluidic application and would like to consider MIC as an SME partner, get in touch with us directly via our website.

