Automated Recirculation Perfusion System
Easy Recirculation
Continuous, unidirectional flow over samples
Automatic Recirculation
No need to switch between reservoirs
Incubator-independent
The cell culture pump replaces the need for a CO2 incubator
Fail-safe mechanism
No more losing your experiment due to clogging

Need a microfluidic SME partner for your Horizon Europe project?
Automated recirculation perfusion system
Our automated recirculation perfusion system is designed to maintain a stable and controlled environment for long-term cell culture. The continuous recirculation allows for real-time monitoring and provides more accurate data on the experimental conditions. Moreover, the recirculation perfusion system reduces the needed reagents compared to single-pass systems, giving you even more control over your experiments.
Moreover, it ensures continuous nutrient supply at constant O2 and CO2 levels without the need for a CO2 incubator. This precision also provides a more physiologically relevant cell microenvironment by sustaining a continuous unidirectional flow through a microfluidic chip laden with cells.
From reservoir 1 to reservoir 2:
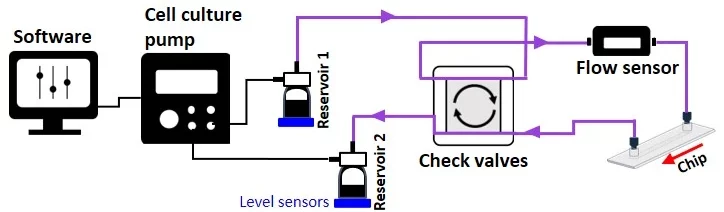
From reservoir 2 to reservoir 1:
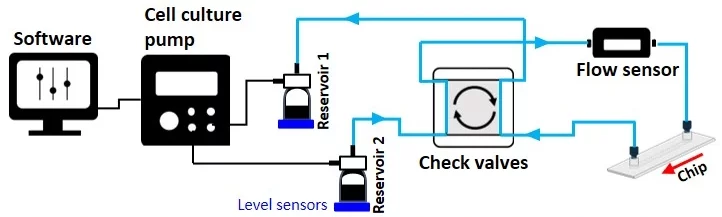
Automated recirculation perfusion system applications
The automated recirculation perfusion system setup includes:

Cell Culture Pump with Gas Control
Keep the pH and oxygen of your media constant outside the incubator
✓ Gas control for incubator-free setup
✓ Stable long-tem flow
✓ Versatile
Cell culture pump

Level Sensors for Microfluidics
Fail-safe instrument for automated recirculation experiments
✓ Easy recirculation
✓ Fail-safe mechanism
✓ Up to 3-week long experiments
✓ Plug-and-play
Level sensors

Check Valve System Pack for Recirculation
Flow control for long-term unidirectional microfluidic experiments
✓ Up to 3 weeks long term perfusion
✓ Setup compatible with incubation
✓ Passive valves, no backflow
✓ Easily stackable for parallelization
Check valves (recirculation bridge)

Microfluidic Flow Rate Sensor
Low flow rate microfluidic flow sensor with automatic clogging detection
✓ Low flow rates
✓ Detect clogging inside the sensor
✓ No drift
Microfluidic flow sensor (eg. Galileo)
Several Schott bottles
Tubings and fittings
Microfluidic chip
User guide
Software (Galileo user interface)
And many more!
The automated recirculation perfusion system setup includes:
Stem Cell Culture
Automated and fail-safe long-term microfluidic cell culture system.
✓ Highly controlled microenvironment
✓ Fail-safe mechanism
✓ Automated sequences
Stem Cell Culture
Microfluidic Spheroid Cell Culture
Plug-and-play instrument pack for automated scaffold-free 3d cell culture technique.
✓ Multiple Parallel culture of Spheroids
✓ Automated Spheroid Perfusion
✓ More Physiologically Relevant Model
Microfluidic Spheroid Cell Culture
Blood-brain Barrier on Chip
Plug-and-play instrument pack for long term BBB on a chip study
✓ Relevant microenvironment
✓ Automatized organ-on-chip perfusion
✓ Plug-and-play microfluidic platform
Blood-brain Barrier on Chip

Sheer Stress
Cell culture system for shear-sensitive cell lines
✓ Safe Neuron Culture Under Flow
✓ Highly Controlled Microenvironment
✓ Up to 3-week Long Cell Cultures
Sheer Stress
And many more!
Have a look at this review comparing this unidirectional automated recirculation system to unidirectional recirculation with a peristaltic pump.
Check this review comparing different bidirectional and unidirectional recirculation systems.
Automated recirculation perfusion system
With microfluidics it is possible to flow medium continuously over cells in culture. A microfluidic recirculation perfusion system involves fluid’s continuous and controlled circulation through microchannels.
Recirculation perfusion systems
However, these systems don’t detect changes and thus continue functioning as programmed. If one of the reservoirs empties before expected due to biofouling or clogging, air enters the system, damaging the cells and ruining the experiment.
In addition, a recirculation perfusion system requires the medium to be supplied with CO2 inside the chip. This can be done using a gas-permeable material, a CO2-independent medium for short periods (about 2 hours), or a CO2 incubator.
One pack for an automated recirculation perfusion system
We have combined the cell culture pump, check valves, and level sensors in one pack for automated recirculation perfusion.

The cell culture pump can be used in the setup as a pressure controller that consumes less gas than other pressure-driven controllers. Pressuring the media reservoir with the correct gas mix prevents the gas in the media from diffusing into the atmospheric air.
The constant media flow ensures that the correct composition always reaches the cells, even if the rest of the system is not gas-tight.
The cell culture pump allows recirculation outside of a CO2 incubator. In this case, an additional stage-top incubator might be used to keep the temperature stable for long periods of time, allowing for high-quality live cell imaging experiments.

The check valve recirculation bridge keeps the flow unidirectional inside the connected microfluidic chip via four passive check valves that are adjusted to quickly and efficiently transfer fluid between reservoirs.
In the figure below, the small red arrows indicate the direction of flow inside each check valve. Corner number 1 marks the common exit from the bridge.
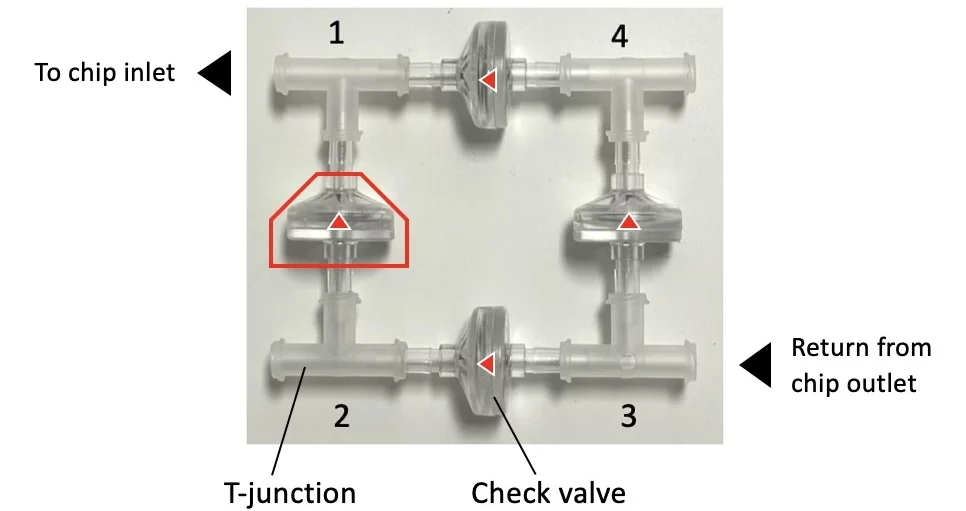

Check this application note for more information on how to assemble the recirculation bridge and set up your experiment.

The level sensors automate the perfusion system based on the media volume level inside reservoirs, ensuring continuous perfusion. In addition, the level sensors introduce a fail-safe mechanism that considers the ever-evolving nature of a perfusion system, such as fouling the tubing with dead cell debris, leakage due to clogging, and other blockage-related issues, such as air entry.
For example, in case of leakage due to extensive clogging, the level sensors will stop the flow, preventing air from being pushed into the system and preserving the sample.
A highly-performant flow sensor for clogging detection
Our engineers have developed a new highly-performant flow sensor, Galileo. This sensor offers <5% flow rate accuracy for sensing ranges from 0.5 to 10,000 µL/min.
Galileo allows for bi-directional flow rate measurement and automatic detection of internal clogging, which will be shown on the small screen of the flow sensor. In case of cross-contaminations or clogging, it’s possible to replace the cartridge for an entirely new flow path. The Galileo flow sensor is a key component that ensures the system’s accuracy and reliability.

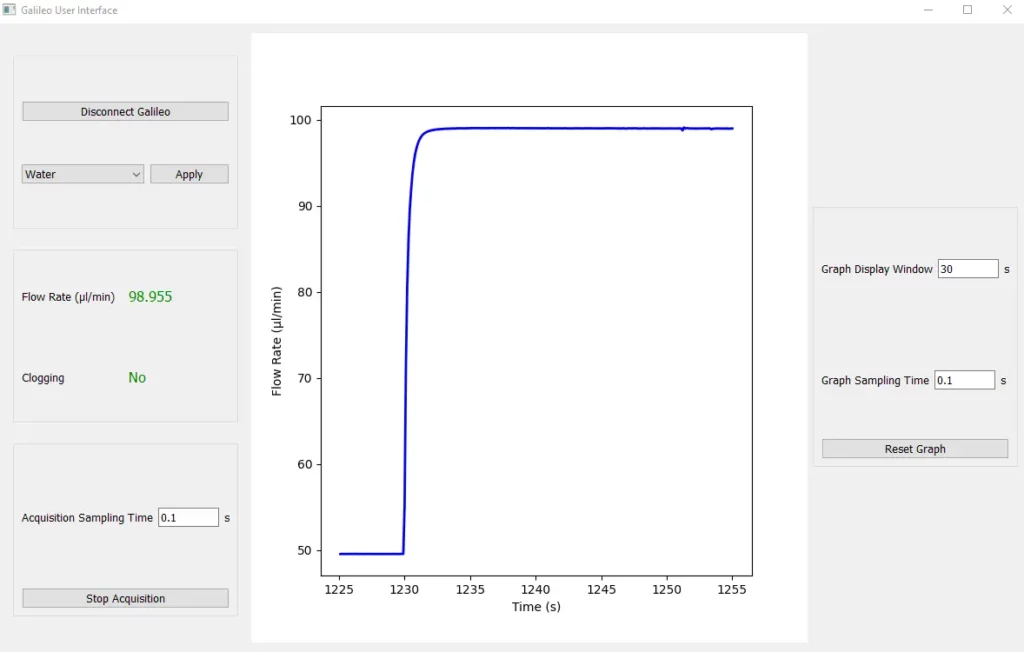

Check the whole video here.
Level sensor technical specifications
The following table summarizes the main specifications of the level sensors:
| Components | Specifications |
|---|---|
| Dimensions (mm) | Sensing area: 2 x 1 cm |
| Material | Plastic |
| Reservoir compatibility | 100 mL Schott bottle |
| Control | Cell culture pump |
Cell culture pump technical specifications
The cell culture pump can come with 1 to 4 channels. Each channel connects to a flow sensor with a feedback loop between pressure and flow rate. Each channel has the following specifications (tested with a set pressure value of 2 bar):
| Characteristics | Specifications |
|---|---|
| Accuracy | -27.75 mbar |
| Air consumption | 0.24 L/min |
| Response time | 140 ms |
| Settling time | 2750 ms |
| Overshoot | 0.12 mbar |
Customize your pack
All the instruments are controlled by the same software, allowing workflow automation and easy integration in your program with free available libraries.
Frequently asked questions
Is the recirculation perfusion system compatible with reservoirs other than bottles?
The current version is compatible with 100 mL bottles, but other options, such as for well-plates, are being developed. Don’t hesitate to contact us if you need a different reservoir type.
Does the cell culture pump keep the gas sterile?
To guarantee the sterility of the used gases, we advise adding a small disposable filter at the gas inlet of the reservoir.
Which microfluidic chips can be connected to the cell culture pump?
The cell culture pump can be connected to any microfluidic chip using the correct connectors.
Can the cell culture pump be placed inside the CO2 incubator?
The cell culture pump is intended to replace the need for a CO2 incubator.
What is the maximum flow rate that can be applied?
The system works well with the range of 0-5ml/min.





Funding and Support
The LIFESAVER project, funded by the European Union’s H2020-LC-GD-2020-3, grant agreement No. 101036702 (LIFESAVER), helped develop the level sensors.
The Tumor-LN-oC project, funded by the European Union’s H2020-NMBP-TR-IND-2020 grant agreement No. 953234 (Tumor-LN-oC), helped develop the cell culture pump.
The ALTERNATIVE project, funded by the European Union’s H2020-LC-GD-2020-3 grant agreement No. 101037090, helped develop the check valve system.
The Galileo project, funded by the European Union’s Horizon research and innovation program under HORIZON-EIC-2022-TRANSITION-01 grant agreement No. 101113098 (GALILEO), helped develop the Galileo flow sensor.
Products & Associated Accessories
FAQ - Automated Recirculation Perfusion System
What then is meant by automatic recirculation? Is the flow bidirectional?
Recirculation is automatic; it does not require manual tubing replacement or reservoir changes during the experiment. The arrangement exchanges fluid between two reservoirs holding a unidirectional flow that is constant yet continuously flowing through the microfluidic chip. In other words: the reservoirs interchange functions, even though the flow is in the same direction as perceived by the chip, which is important to systems that are sensitive to shear and to prevent a periodic mechanical disturbance.
What is so important about the unidirectional flow of organ-on-chip or sensitive cell lines?
Since many biological readouts do not simply rely on average flow, they also assess the stability of flow over time. Unidirectional perfusion eliminates periodic reversals that can be used to detach cells, remix gradients, or generate artificial mechanical stimuli that lack physiological significance. When you are doing endothelial models, neuron culture under flow and barrier tissues, such stability is the difference between publishable biology and inexplicable variability.
What is the effect of this in the absence of a CO2 incubator? Is CO 2 regulation required as a matter of (nominally) necessity by pH?
Yes, normally, particularly with media buffered with bicarbonate. In this case, the cell culture pump has gas control so that the media in the reservoir can be held at the desired CO2/O2 conditions, not within a classical incubator. In practice, it is what causes experiments that are independent of the incubator to be realistic, especially when you need access to live imaging or you simply want your entire fluidic system to be available on the bench. The last component is temperature; many teams use this with a stage-top or local thermal control when making long imaging times.
What then is the “fail-safe mechanism?
The fail-safe logic is based on level sensors that measure the reservoir volume and deactivate the flow if the system becomes unsafe (e.g., due to leakage or extreme clogging). It does not matter to be wise to be conservative: to prevent air from being blown into the circuit. That is a remarkably large quality-of-life improvement over the long term, since it avoids the kind of silent failure you only discover the following morning.
How long would experiments effectively last with this setup?
Design goal: It can support up to ~3-week-long experiments in automated recirculation/perfusion, provided you have the rest of your biology (media stability, contamination control, chip design, tubing choice) configured to support them. It is in this window of weeks and not days that recirculation finds appeal: you maintain the system stable but save on reagent/media usage than using single-pass techniques.
What are its supported flow rates?
In normal operation, the system can operate in the 0 to 5 mL/min range. Individually, the flow sensor component (Galileo) has low-to-high microfluidic working capabilities, reported sensing ranges (between 0.5 and 10,000 uL/min), and less than 5% accuracy in flow-rate measurements. That comes in handy when you have a concern over what is actually being received by the chip (not necessarily what you believe is being received).
What will be contained in the pack and what am I still required to supply?
The pack idea is meant to be a pragmatic one:
- Gas control cell culture pump (stable long-term flow and gas conditioning incubator-free)
- Level sensors (+ fail-safe stopping behavior)
- Passive (keep flow in one direction, allow it to backflush; may be stacked together to be used in parallel) check valve bridge.
- Flow-rate sensor (Galileo) + software (feedback, clogging detection, monitoring).
You continue to include the biology-specific components: your microfluidic chip, cell-handling workflow and the kind of tubing/fittings that you would like (though standard tubings, fittings and Schott bottles are included in the standard setup package).
How do you keep the gas path sterile if you’re controlling CO₂/O₂ into reservoirs?
The simplest method is suggested: a small, disposable, sterile filter is suggested at the gas inlet into the reservoir. It is one of those facts that seems to be optional, until the time that you have lost a multi-week culture to something that was not created in your cells at all.
Which microfluidic chips are compatible?
Mechanically, it is wide: the pump can be connected to any microfluidic chip, provided it has the connectors and fittings required to match the ports or pressure/flow specifications of your chip. The more significant compatibility question is generally biological and fluidic: the dimensions of channels, bubble control, adsorption, shear sensitivity, and your imaging and sampling intentions.
Where does a microfluidic SME partner actually fit in Horizon Europe proposal?
A good SME partner in Horizon Europe consortia can minimize technical risk (prototype design, iterative microfluidic design, system integration), accelerate validation (automation, consistent perfusion control, sensor-guided sensing), and ensure exploitation is credible (manufacturability, technology transfer, deliverable-grade prototypes).
The areas where MIC has the best positioning usually include: microfluidic setup design, perfusion, automation/software integration, reporting, milestones, and reproducibility. Another consistent trend is that proposals that include a working microfluidic SME with proven delivery potential are more persuasive for implementation and impact.

