Cell Culture PH Control without CO2 Incubator
pH control in dynamic cell culture independent of a CO2 incubator.
Be independent of the CO2 incubator
Control pH of the media on the bench
Automated cell culture
Less manual work and more accuracy for your experiments
Plug-and-play platform
Beginner friendly pack with detailed user guide

Need a microfluidic SME partner for your Horizon Europe project?
pH control without a CO2 incubator for dynamic cell culture
Aware of the importance of pH control in dynamic cell cultures, this pH control without a CO2 incubator pack was assembled to closely maintain cell culture pH in line and independently of the CO2 incubator by maintaining the gas composition used to pressurise the reservoirs. Assemble your setup at the bench or on the microscope stage for better data gathering.
This pack for pH control without a CO2 incubator is user-friendly, customizable, and automatable perfusion pack. You can add your desired gas composition, such as 5% CO2/95% air, through a pre-mixed bottle connected to the pump. The pump, in turn, will ensure this composition remains constant throughout your experiment. The Stage Top Incubator keeps the temperature, forgoing the need to use a CO2 incubator.

A typical pack contains:
Flow sensor (Galileo, MIC)
Software (Galileo user interface)
Cell culture pump
Eppendorfs or falcon reservoirs
Recirculation bridge
A microfluidic chip, if needed
User-friendly guides
This pack can also be combined with other microfluidic steps following the dynamic cell culture, such as drug sequential injection.
pH control without a CO2 incubator for dynamic cell culture

The homeostasis of pH is finely balanced in the body by a bicarbonate buffering system, creating a dynamic equilibrium between carbonic acid and carbonate, which, in turn, is regulated by the lungs and kidneys. The same buffering system is usually applied to cell cultures with a CO2 incubator that keeps CO2 at a predefined concentration.
Phenol red is the most common indicator of pH in cell culture media, and it is usually used as a visual cue of the culture’s development.

The microenvironment changes become more relevant when the cell culture is miniaturized to take advantage of the benefits of organ-on-a-chip technology. Even if slight variations in pH, e.g., 7.4±0.3, are tolerated in traditional cell culture, the pH changes faster in microfluidic cell culture due to the much higher cell volume to medium volume ratio, resulting in a more pronounced effect on cell viability. Thus, closer monitoring of crucial environmental parameters becomes more critical [1].
References
1. Lu, C. & Verbridge, S. S. Microfluidic methods for molecular biology. Microfluid. Methods Mol. Biol. 1–376 (2016). doi:10.1007/978-3-319-30019-1
pH control without a CO2 incubator
Most researchers rely on Phenol Red to assess the pH of cell culture and cell experiments. However, the same shade of Phenol Red can represent an entire pH point, which can be problematic since the dimensions of most organ-on-chip devices make small pH changes that significantly affect cells.

Due to the bicarbonate buffering system commonly employed in cell culture media, the pH of the cell culture is also tightly linked to the concentration of CO2 in the atmosphere. The graph below illustrates the significant pH change of DMEM when exposed to different atmospheric concentrations of CO2.
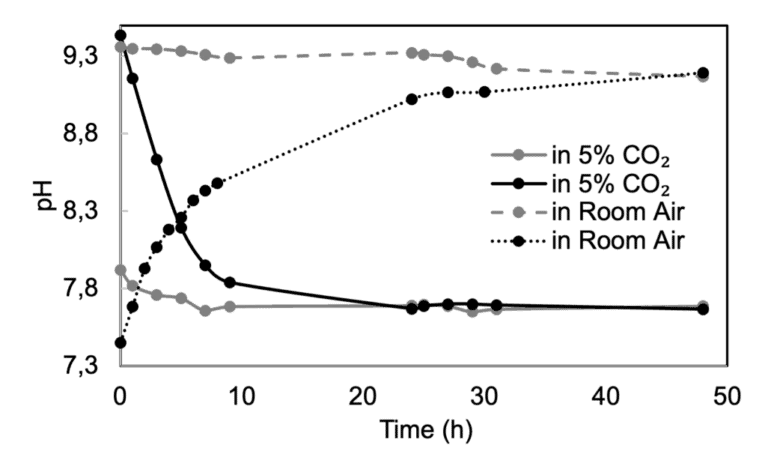
This reliance on CO2 renders most dynamic cell culture setups dependent on the CO2 incubator, limiting the technology’s potential. For example, image gathering on a microscope requires unplugging the chip from the setup, which can cause contamination and air bubbles.
Live cell imaging is also limited as the device cannot remain outside ideal conditions, requiring specialized equipment. These limitations call for a way to culture cells dynamically independent of the CO2 incubator.
Customize your pack
This pack can be modified depending on your cell line (human, mammalian, plant, primary…) or your experiment conditions.
If you’re unsure about the instrument choices and settings best suited for your application, contact one of our experts!
Bubbles can be a problem for cells; the instrument pack can contain a bubble remover to tackle this issue efficiently.
Our experts can answer any questions about this pH control without an incubator pack and help you find out how it can match your specifications.
Frequently asked questions
Which gases can the cell culture pump use?
The cell culture pump can use any non-corrosive gas mixture.
Does the cell culture pump keep the gas sterile?
To guarantee the sterility of the used gases, we advise adding a small disposable filter at the gas inlet of the reservoir.
Do the reservoirs of media need to be kept at 37°C?
No, the reservoirs can be kept at room temperature. The Stage Top Incubator was designed to ensure that the media reaches the cells at the desired temperature regardless of the temperature of the reservoirs.






Funding and Support
The results of the Protomet, ALTERNATIVE, LIFESAVER, and Tumor-LN-oC projects helped develop this pH control pack, with funding from the European Union’s Horizon 2020 MSCA-ITN under grant agreement No 813873 (ProtoMet), the European Union under H2020-LC-GD-2020-3, grant agreement No. 101036702 (ALTERNATIVE project), the European Union under H2020-LC-GD-2020-3, grant agreement No. 101037090 (LIFESAVER project),
Products & Associated Accessories
FAQ - Microfluidic pH control without a CO₂ Incubator
What is the Cell Culture pH Control Without CO2 Incubator Pack?
This pack is a complete microfluidic system that provides a stable pH in dynamic cell cultures without the need for a traditional CO2 incubator. It can reproduce the buffering conditions of a CO2 incubator by regulating the composition of the gas the researcher wishes to use to pressurize the media reservoirs, and can be used to directly run and monitor cell culture experiments on the bench or even on the stage of a microscope.
What is so important about pH regulation in microfluidic cell culture?
Although modest pH differences, e.g., 7.4 ±0.3, may be permitted in conventional cell culture, in microfluidic cell culture, pH changes occur more rapidly due to the much greater cell-to-medium volume ratio, with a more pronounced effect on cell viability. This implies that the safety margin used by the researchers at the macroscale does not directly apply to chip-based systems. Therefore, closer, more continuous pH monitoring and control are not a convenience but a necessity.
Why is the use of a CO2 incubator a drawback of dynamic cell culture?
Most dynamic cell culture systems are also reliant on the incubator, making the technology susceptible to failure. For example, to collect images under a microscope, one must remove the chip from the system, which may introduce contamination and air bubbles. Another limitation of live cell imaging is that the machine cannot remain in optimal conditions for extended periods, and specialized equipment is usually required. This pack eliminates such dependency, and so experiments can proceed endlessly at the bench or on the microscope stage.
What is the mechanism of pH maintenance of the pack without the CO2 incubator?
The pack balances pH by varying the gas mixture used to pressurize the media reservoirs. The cell culture pump is connected to a pre-mixed gas bottle containing 5% CO2 and 95% air, so that this ratio is maintained throughout the experiment. Because the bicarbonate buffering system in standard cell culture media is directly linked to the surrounding CO2 concentration, stabilizing the gas atmosphere over the media is the same step as stabilizing pH, which is typically performed by the incubator.
What are the gases that the cell culture pump can use?
The cell culture pump can be supplied with a non-corrosive gas mixture. This provides the researcher a lot of flexibility to recreate various physiological or experimental atmospheres – standard 5% CO2 / 95% air to culture mammalian cells, a low oxygen environment to study hypoxia, and other specifications that may be necessary to the particular cell line or experimental model.
What is the method of gas sterility during the experiment?
A small disposable filter placed at the gas entry point into the reservoir will ensure the sterility of the gases entering the reservoir. This simple precaution ensures that no airborne contaminants are introduced into the culture medium or the pressurizing gas, and this is especially true in long-term experiments, where sterility is difficult to achieve otherwise.
Should the media reservoirs be kept warm?
No, the reservoirs may be kept at room temperature. The Stage Top Incubator is designed so that the media reaches the cells at the correct temperature, independent of the reservoir temperature. The heating is applied along the fluid pathway; thus, rather than the entire media volume being pre-warmed, the heating is confined to the fluid pathway. This makes arranging the bench much easier and reduces the need for thermal control.
Why is Phenol Red not enough to monitor pH in microfluidic systems?
The most widely used pH indicator in cell culture media is Phenol Red, but the same shade can indicate a full pH unit. Small pH shifts that are visually analogous can have a significant impact on cells in most organ-on-chip devices. The internal tests revealed that DMEM placed in the CO2 incubator may shift to the 9.3-9.5 range, and to the 7.5-7.7 range when exposed to air, with significant changes occurring in the first 10 hours of exposure. Color presentation is insufficient for the accuracy needed in microfluidic cell culture.
What does a standard pack consist of, and can it be customized?
A typical package comprises a flow sensor, control software, a cell culture pump, Eppendorf or Falcon reservoirs, a recirculation bridge, a microfluidic chip (if needed), and user-friendly manuals. The pack can be fully configured for the cell line (e.g., human, mammalian, plant, or primary cells) and the experimental conditions. Several additions are optional, e.g., a bubble remover to prevent air exposure of the cells, and more complex workflows may be combined with the pack using other microfluidic additions, such as drug sequential injection.
Which cell culture applications are supported by this pack?
The pack has been developed for use in any dynamic cell culture where physiological pH is required, outside a CO2 incubator. This covers organ-on-a-chip tests, live-cell microscopy imaging, long-term perfusion cultures, and research on drug delivery or environmental stimuli. Since the system can be automated and has been reported to be novice-friendly with user manuals, it can be used by microfluidic researchers as well as new and inexperienced chip-based cell culture users who require a solid, plug-and-play system.

