Microfluidic droplet generation to produce artificial cells: ACDC
Author
Christa Ivanova, PhD
Publication Date
January 08, 2019
Status
Keywords
artificial cells
monodisperse emulsions
biomimetic systems
drug screening
protein function
in-vitro synthetic biology
droplet generation
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
Artificial cells with distributed cores to decipher protein function: introduction
ACDC project aims to develop a new technology for creating simple artificial cells made of liquid-based capsules. These capsules will contain interactive and multifunctional cores (sensing, actuating, reporting, energization, and barcoding functions) and communicate micro-laboratories.
This new technology reproducing living cells will help identify new targets for future drugs and conduct drug screening against the identified proteins.
Microfluidics allows the generation of specific-size droplets and monodisperse emulsions in a very controlled way. This technology is used in the ACDC project to produce artificially engineered cells.
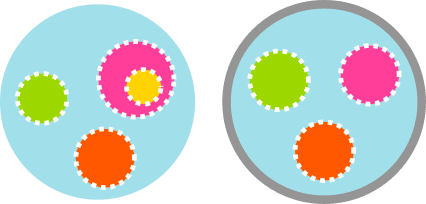
Artificial cells with distributed cores to decipher protein function: project description
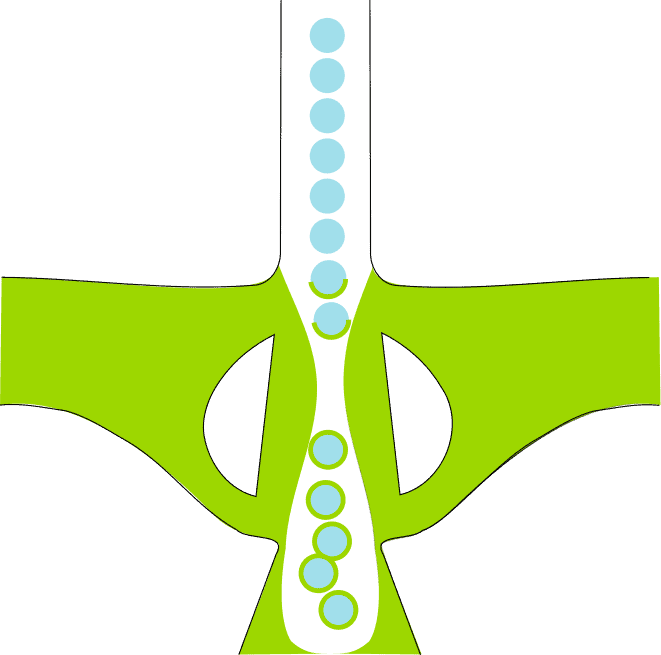
The capsules will be made using microfluidics technologies, particularly droplet generators based either on flow-focusing geometries to obtain the cores of the future capsule or on an innovative bat-wing junction to encapsulate these cores into the capsule.
The new generation of microfluidic devices designed for this purpose will be made by 3D printing, allowing the construction of 3D structures to produce artificial cells more effectively.
In the ACDC consortium of 6 European partners, we will design the chips and optimize fluid handling to produce cells.
Elveflow microfluidic flow control system will help us precisely adjust each liquid’s volume (matrice and reagents) over an elongated duration to obtain reproducible capsules and droplets. We will also perform on-chip-specific DNA labeling of the celand as drug screening.
Related content & results from this project
As a result of the ACDC project, we developed the artificial cell droplet pack.
The project also helped develop other paks:
Download the MIC Horizon Europe 2026/2027 Calls Calendar:


Funding and Support
This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 824060 (ACDC project).
Check our Projects
FAQ – Microfluidic droplet generation to produce artificial cells: ACDC
What is the reason microfluidics is being used on "artificial cells with distributed cores" as opposed to bulk emulsification?
Droplet microfluidics provides deterministic control since it is a problem that is easily controllable. You can produce droplets of any size and very monodisperse emulsions, select the rate of flow to couple reagents on demand, and achieve long-run stability. That is necessary when you have to do reproducible capsules to screen and perform protein-function assays.
What are the geometries of the construction of these capsules?
Two mutually suitable strategies. This is first done using classical flow-focusing junctions to shape and fill the individual cores. Second, a new type of junction, the so-called bat-wing junction, is used to enclose those cores in the last capsule. The two-stage architecture minimizes variability and loads multi-cores, which is tractable.
Are microfluidic devices traditionally made?
Not exclusively. The project used 3D printing to develop real 3D microstructures for assembling capsules. That option speeds up iteration (by using smaller mask steps) and allows you to model complex junctions that are cumbersome to fabricate in a planar process.
What was the scientific application of the project?
Exploring protein-scale functionality and, downstream, drug discovery. These artificial cells are used to discover new protein targets by mimicking important cellular features, such as compartmentalization, controllable reactions, and readouts and screen compounds against these artificial cells under tightly controlled conditions.
What was the contribution of the Microfluidics Innovation Center (MIC)?
MIC developed microfluidic chips, optimised fluid handling to stabilise multiphase flows, and conducted long-term experiments with well-controlled volumes to obtain reproducible droplets and capsules. The team also introduced on-chip labeling steps and established screening-ready workflows, taking the team from the idea of the device to a usable protocol.
What instruments and control strategies played a vital role with regard to reproducibility?
Precise flow control to maintain tight tolerances over extended periods for each liquid, e.g., in pressure-driven systems and reagent applications. This, together with feedback on droplet formation, helps reduce drift, maintain monodispersibility, and enable repeatable multi-core encapsulation.
Is this different from liposomes or single-compartment vesicles?
Classical vesicles provide one reaction volume. In this case, two or more cores in a single capsule permit side-by-side execution of orthogonal functions, e.g., sensing in one, energy in another, and reporting in a third, all with exchanged signals. The distributed design has the advantage of easing the choreography of complex assays.
Could these artificial cells be modified for my assay?
Yes. At the chip level, core count, loading sequence, reagent stoichiometry, and shell properties can be programmed. When your readout is based on nucleic-acid tagging or fluorescent reporters, the on-chip DNA-labeling capacity reduces the road to a screening-grade protocol.
What is the rationale of including MIC in a Horizon Europe consortium on such work?
Because an established microfluidics SME provides de-risked engineering: chip design, flow-control integration, and prototype-to-prototype transfer. MIC has a regular presence in EU consortia, has prototypes released to the market rapidly, and is a co-writer of proposals; our experience is that consortia in which MIC participated, with a practical microfluidics SME, have higher success rates of implementation than the formal baseline success rates of proposals, two to one in our case.