Cell Perfusion with Oxygen Control
Control the oxygen of your dynamic cell culture continuously
Plug in your pre-mixed gas bottle
Pressurize your media with the desired oxygen concentration
Versatile
Compact and portable pump
Stable long-term flow
Stable flow in the physiological profiles of your choice

Need a microfluidic SME partner for your Horizon Europe project?
Oxygen control setup
We have assembled all the components to perform successful perfusion with continuous O2 control. The setup displayed below is meant to continuously and stably perfuse media in a highly controlled microenvironment: a pre-mixed gas bottle is plugged into the cell culture pump, pressurizing the reservoirs with the correct mix of gasses for your experiment; temperature is kept by a stage top incubator that allows for long-term live cell imaging with ease.
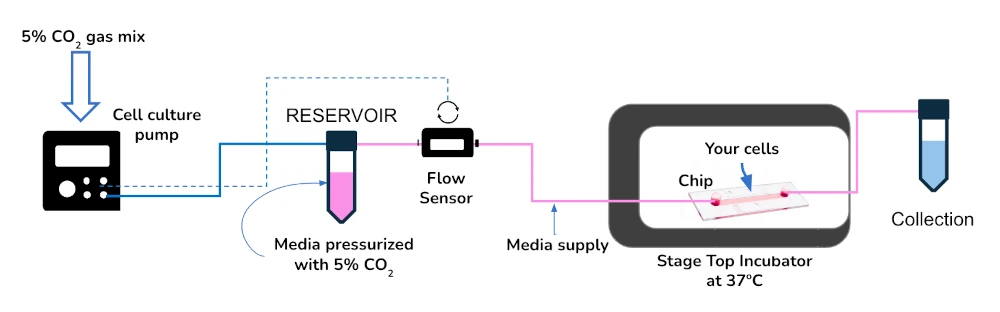
The cell culture oxygen pack contains:
Flow sensor (Galileo, MIC)
Software (Galileo user interface)
Cell culture pump
Stage top incubator
Microfluidic chip (application-dependent; suggestion ibidi µ-Slide I Luer)
Fittings, tubings, luers
Reservoirs
User guides for instruments
This instrument can also be adapted to our automated cell culture platform.
For applications requiring fluid injection to test the effects of drugs, for example, a rotary valve or a check valve recirculation bridge can be added to the setup. Check out our automated recirculation system and this application note that explains how to assemble the recirculation bridge.
Oxygen levels in dynamic cell culture
Adequate microenvironment parameters are crucial for a thriving cell culture. Oxygen in the cell culture is one of the parameters that must be surveyed to ensure healthy cell growth and division.
However, it is rarely monitored in traditional cell culture flasks since it is assumed that the oxygen from the atmosphere will diffuse into the media at sufficient rates.
On the other hand, dynamic cell cultures are closed-loop systems in which the changes in concentration of essential parameters, such as oxygen, are more pronounced due to the minute volumes. Therefore, they require closer monitoring and surveillance.
Applications
Red blood cell studies
Oxygen supply for gas impermeable chips
Effects of oxygen concentration on cell viability and gene expression
And many more!
Chip compatibilities

PMMA device station
From mold development to high-resolution PMMA chip fabrication
✓ Fast process
✓ Multiple applications
✓ Simple and innovative setup
PMMA
Homemade devices, PDMS
Synvivo
Microfluidic ChipShop
And many more!
Oxygen control pack specifications
The Oxygen control pack contains a set of instruments for continuous cell culture perfusion and monitoring. The cell culture pump controls the fluid flow.
The table summarizes the main specifications of the Oxygen control system.
| Components | Technical Specifications |
|---|---|
| Cell culture pump | 4 Channels (0 to 2000 mbar) |
| Microfluidic flow sensor (eg. Galileo) | Range from 0.5 to 10,000 µL/min |
| Cell culturing chamber | Microfluidic chips (2.55 x 7.55 cm) |
| Stage top incubator | 0 to 70oC; k-frame bottom |
Customize your pack
Our instruments can be added to highly customizable setups, so you can take advantage of the extra flexibility to adapt them depending on your specific needs. Our microfluidic specialists will advise you on the best instruments and accessories depending on your needs and will accompany you during the setup of the microfluidic platform.
Our instruments are compatible with standard commercialized chips from different brands.
Frequently asked questions
Is the setup sterilizable?
The reservoirs and tubings are disposable but can be sanitized with a cleaning protocol. The flow sensors can also be sanitized with the same protocol. Connectors can be autoclaved.
Can I do anaerobic/hypoxic experiments with this setup?
For example, you can plug a nitrogen bottle into the cell culture pump to pressurize a deoxygenated media. However, the gas permeability of the tubing and the gas tightness of the fluidic system connections must be considered to ensure no oxygen permeates into the system during the experiment.
Can the pump be placed in the CO2 incubator?
The gas control pump should be kept outside the CO2 incubator. The flow sensors can be placed inside the incubator.
Can it measure the oxygen concentration in the media?
Currently, the pump can only control the gas composition based on the pre-mixed gas input supplied. It is advisable to add oxygen sensors to the microfluidic circuit or chip if there’s a need to measure the precise concentration of oxygen.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:



Funding and Support
LIFESAVER and Tumor-LN-oC projects helped develop this pack. These projects are funded by the European Union’s H2020-LC-GD-2020-3 and H2020-NMBP-TR-IND-2020, grant agreements No. 101036702 (LIFESAVER), and No. 953234 (Tumor-LN-oC).
Products & Associated Accessories
FAQ - Cell Perfusion with Oxygen Control
What is the goal of the Cell Perfusion with Oxygen Control setup?
The goal of this setup is to allow a constant flow of cell culture media in a tightly controlled micro-environment, particularly to maintain a precise control of oxygen levels during a dynamic cell culture experiment.
What components are included in the Oxygen Control Pack?
The cell culture oxygen pack contains:
- Flow sensor (Galileo)
- Software (Galileo user interface)
- Cell culture pump
- Stage top incubator
- Fittings, tubings & luers
- Reservoirs
- Microfluidic chip (application-dependent; suggestion ibidi µ-Slide I Luer)
- User guides for instruments
How does the oxygen control actually work?
The cell culture pump is directly connected to a pre-mixed gas container. The control of dissolved oxygen to the cell culture is determined by how the pump pressurizes the cell culture media reservoirs with a particular gas mixture. For long-term live cell imaging, a constant temperature is maintained at a stage-top incubator. The pump does not have a function to mix gases; it is determined by which gas input container is chosen.
What flow rates can the system handle?
The flow rates of this system can handle most physiological perfusion rates required in organ-on-a-chip and dynamic cell culture studies, which fall within a range of 0.5 to 10,000 µL/min of the Galileo flow sensor.
Can the setup be sterilised?
Although the tubings and reservoirs are for single use only, they may be sterilised using a specific cleaning protocol. The same protocol may be used for sterilising the flow sensors as well. Autoclaving may be used for sterilising the connectors.
Can you carry out anaerobic or hypoxic studies using this setup?
Yes. The system may be used to pressurise deoxygenated fluids by tying a nitrogen bottle to the pump. Two important aspects have to be carefully attended to in this regard. The gas permeability of the tubing and gas-tightness of all the fluidic interfaces have to be ensured. The presence of any oxygen in the system due to tubing or connector interfaces may spoil the hypoxic environment. Hence, gas-impermeable tubing is recommended for this purpose.
Where should the pump be placed in relation to the incubator?
The CO2 incubator should not be used for housing the gas control pump. Only the flow sensors may be placed inside the incubator. This is due to the gas supply interfaces and the electronics of the pump.
Is it possible for the system to detect the amount of oxygen dissolved in the medium?
Not by itself. The pump does not have an oxygen sensor. It simply mixes the gas composition based on the input received. If you need to know the concentration of dissolved oxygen in real time, you should add an oxygen sensor to the system. You can choose an optical fibre or electrochemical sensor and add it to the microfluidic circuit or chip.
What about adding fluid switching or drug/reagent injection to the system?
Yes. You can add a rotary valve or a recirculation bridge with a check valve to inject medications or test substances into the system. Also offers an automated recirculation system that is compatible with this system.
Is it possible for the system to be adapted for automated platforms?
Yes. You can integrate the Oxygen Control Pack with automated cell culture platform. This will increase the throughput and automate the process. The configuration will depend on what is required.

