Liposome Synthesis
Synthesis of liposomes using microfluidics
Efficient liposome nanoparticle synthesis
High throughput, reproducibility and small size distribution
Easy system implementation
Unbox, set up and start your experiments right away
Straightforward scale up
Produce your liposomes at any volume (µL to L)

Need a microfluidic SME partner for your Horizon Europe project?
Liposome nanoparticle synthesis
The liposome nanoparticle synthesis pack can be customized to fit your specific needs. It contains at least two pumping channels to flow your two solutions – generally water and solvent – needed to perform the liposome synthesis inside your mixing chip. Liposomes can be synthesized using this liposome synthesis pack!
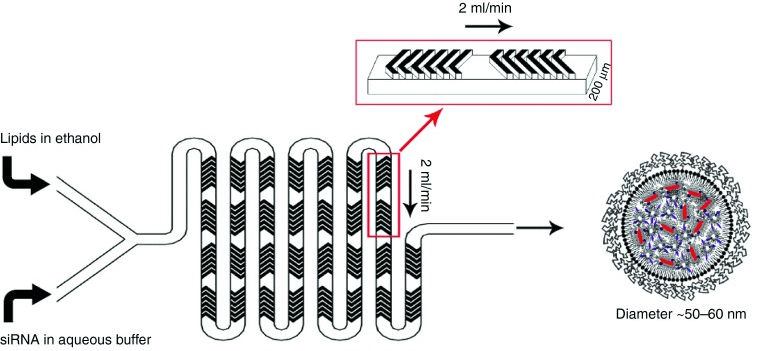
The microfluidic chip where the liposome synthesis occurs is a staggered herringbone chip with two inlets and induces chaotic mixing even at a microfluidic scale. The first inlet is used to provide the liposomes in ethanol, and the second one is used to bring the aqueous solution and possibly a hydrophile load that will be encapsulated inside the newly formed LNP, like siRNA, for example (see the application tab). At lower flow rates, other chips, such as coflowing ones, can be used to optimize your liposome production yield.
The production can be easily scaled up by increasing the volumes and flow rates and parallelizing several micromixers instead of one, thus increasing the system’s overall throughput while maintaining monodispersity and yield.
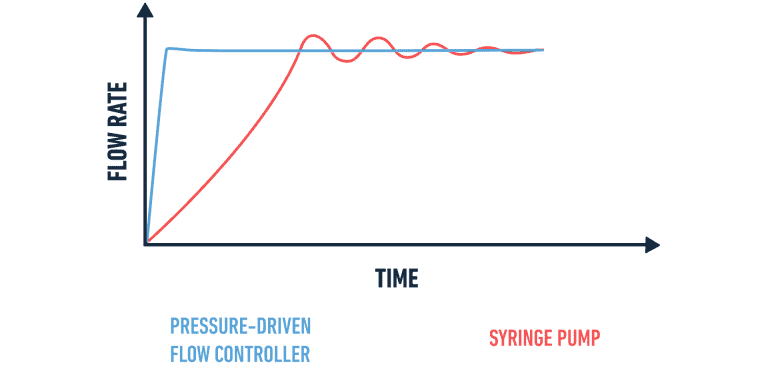
The stability and the speed of the reaction directly depend on the flow rates of each fluid in the microfluidic channel. The flow controller creates the flow, and the flow rates are measured and regulated thanks to a flow sensor that makes a feedback loop for continuous flow control.
The combination of these instruments is the fastest and most precise microfluidic flow control available on the market, guaranteeing the best possible liposome monodispersity.
Pressure-driven flow control systems are more suited for liposome synthesis than peristaltic or syringe pumps because they are pulseless and can reach the desired flow rate in the chip way faster than other systems.
Liposome synthesis pack setup
This plug-and-play instrument LPN synthesis pack ensures a good compatibility between the different instruments, allowing the experiment to be started right away. The whole system is controlled by a single software and can be used for a wide variety of applications.
Contact our experts for a detailed protocol for microfluidic liposome synthesis.
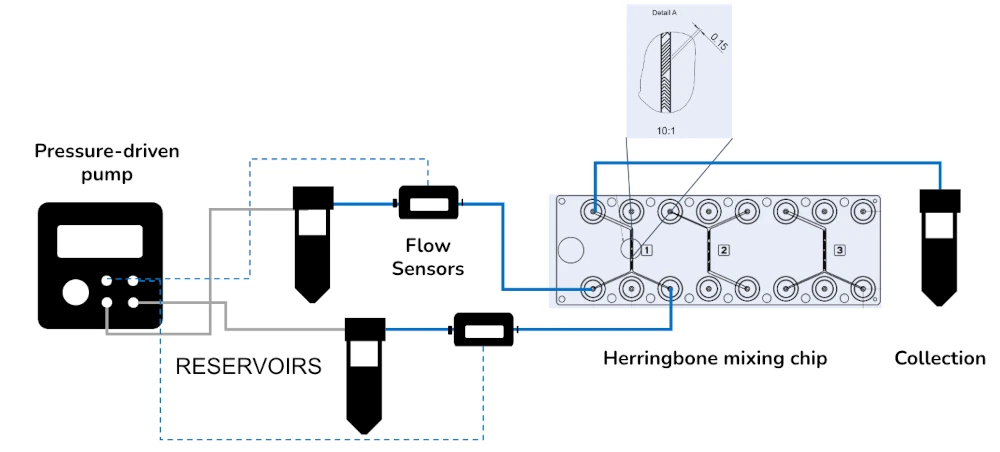
A typical liposome pack contains:
Flow sensor (Galileo, MIC)
Software (Galileo user interface)
Pressure-driven pump
Reservoirs
Chemical solutions to perform liposome synthesis if required: lipids, surfactants,…
One or several Fluidic 187 Herringbone mixer from microfluidic ChipShop (a flow focusing chip can also be used)
T-junctions/9 manifolds if production parallelizing
All necessary accessories: tubing, connectors, filters, etc…
Liposome synthesis is also possible using a flow-focusing method with a different microfluidic chip.
Microfluidics herringbone liposome nanoprecipitation principle
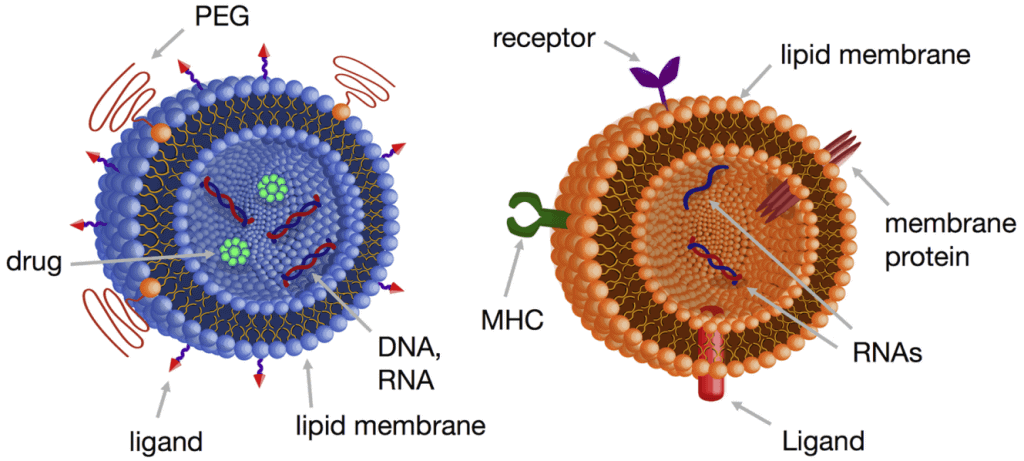
Ribonucleic acid (RNA) is a critical polymeric molecule for gene regulation and expression. RNA interference (RNAi) is a method that silences genes by using sequence-specific small interfering RNA (siRNA).
Based on their mRNA counterpart nucleotide sequences, siRNAs block the production of specific proteins [1-3]. Lipid nanoparticles (LNP) are the most commonly used system for delivering siRNA for therapeutic applications in vivo, such as anti-tumor agents or polyneuropathy treatment [4-5].
BioNTech/Pfizer’s BNT162b2 and Moderna’s mRNA-1273 vaccines also use lipid nanoparticles as vehicles for mRNA delivery into the cytoplasm of host cells, which leads to the production of COVID-19-Neutralizing antibodies [5]. Thus, lipid nanoparticle synthesis has played a critical role in developing COVID-19 vaccines and other nanomedicines and is considered very promising for developing new drug delivery systems [6].
Liposome nanoparticles can be used as cosmetic delivery systems for other applications, like solid lipid nanoparticles (SLN) [7].
The liposome nanoparticle synthesis pack includes staggered herringbone micromixers that create chaotic flows. They are the most commonly used microfluidic chips for LNP synthesis [8]. The structure generates chaotic advection, which can induce rapid mixing at Reynolds numbers smaller than 1 [9], thus allowing a better encapsulation efficiency than bulk methods [10].
References
Whitehead, K., Langer, R. & Anderson, D. Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov 8, 129–138 (2009).
Belliveau N., Huft J., Lin P., Chen S., Leung A., Leaver T., Wild A., Lee J., Taylor R., Tam Y., Hansen C., Cullis P., Microfluidic Synthesis of Highly Potent Limit-size Lipid Nanoparticles for In Vivo Delivery of siRNA, Molecular Therapy – Nucleic Acids, 1, 2012, e37, 2162-2531
Allen, Theresa M., and Pieter R. Cullis. “Drug delivery systems: entering the mainstream.” Science 303.5665 (2004), 1818-1822.
Sasayama Y., Hasegawa M., Taguchi E., Kubota K., Kuboyama T., Naoi T., Yabuuchi H., Shimai N., Asano M., Tokunaga A., Ishii T., Enokizono J., 2019, In vivo activation of PEGylated long circulating lipid nanoparticle to achieve efficient siRNA delivery and target gene knock down in solid tumors. Journal of Controlled Release
Let’s talk about lipid nanoparticles. Nat Rev Mater 6, 99 (2021).
Thi, T.T.H.; Suys, E.J.A.; Lee, J.S.; Nguyen, D.H.; Park, K.D.; Truong, N.P. Lipid-Based Nanoparticles in the Clinic and Clinical Trials: From Cancer Nanomedicine to COVID-19 Vaccines. Vaccines 2021, 9, 359
Sylvia A Wissing; Rainer H Müller (2003). Cosmetic applications for solid lipid nanoparticles (SLN), 254(1), 65–68.
Maeki M., Kimura N., Sato Y., Harashima H., Tokeshi M., 2018, Advances in microfluidics for lipid nanoparticles and extracellular vesicles and applications in drug delivery systems. Advanced Drug Delivery Reviews
N-T. Nguyen, Z. Wu, Micromixers-a review, J. Micromech. Microeng. 15, 2005, R1-R16.
Leung A., Hafez I., Baoukina S., Belliveau S., Zhigaltsev I., Afshinmanesh E., Tieleman P., Hansen C., Hope M., and Cullis P., The Journal of Physical Chemistry C 2012 116 (34), 18440-18450
Why use microfluidics for liposome nanoparticle synthesis?
Liposome nanoparticles with a mean diameter between 20 nm and 100 nm have been synthesized using a system equivalent to the one that can be assembled using this pack [1-2]. Liposomes have also been synthesized with similar dimensions with improved reproducibility [3].
Microfluidic nanoprecipitation enables a good throughput, monodispersity with tunable particle size, and, generally, a better control of the nanoparticle synthesis. The encapsulation efficiency is also higher at the microfluidic scale.
One of the other substantial advantages of a microfluidic platform compared to a bulk method for liposome synthesis is that this process can be easily included as a single step in a more complex operation by adding more microfluidic instruments and chips.
References
Belliveau N., Huft J., Lin P., Chen S., Leung A., Leaver T., Wild A., Lee J., Taylor R., Tam Y., Hansen C., Cullis P., Microfluidic Synthesis of Highly Potent Limit-size Lipid Nanoparticles for In Vivo Delivery of siRNA, Molecular Therapy – Nucleic Acids, 1, 2012, e37, 2162-2531.
Rungta, Ravi L., et al. “Lipid nanoparticle delivery of siRNA to silence neuronal gene expression in the brain.” Molecular therapy. Nucleic acids 2.12 (2013).
Kotouček, J., Hubatka, F., Mašek, J. et al. Preparation of nanoliposomes by microfluidic mixing in herring-bone channel and the role of membrane fluidity in liposomes formation. Sci Rep 10, 5595 (2020).
Customize your microfluidics liposome nanoparticle production
Contact our experts to answer any questions about this liposome nanoparticle synthesis pack and how it can match your specifications!
Frequently asked questions
How can we help your experiment?
This pack is in beta testing phase. So, although the instruments are not fully industrialized, we can provide extensive support as part of our beta testing program. Get in touch to see if you are eligible.
Can a pack be customized based on my specific application?
Yes! Our experts will establish which instruments are best suited for your application, such as the type of flow sensor or the number of flow controller channels you need to perform your experiment. Contact us using the “talk to our experts” green button above.
Can I buy individual instruments?
Our instruments are in beta testing phase and can be tested as a pack or individually, so get in contact with our team to know how our beta testing program works.
Products & Associated Accessories
FAQ - Liposome Synthesis Pack
What is the Liposome Nanoparticle Synthesis Pack and what?
Liposome Nanoparticle Synthesis Pack is a microfluidic home-assembly that synthesizes lipid nanoparticles (LNPs) with high reproducibility, small size distribution, and tuned particle sizes. It incorporates a pressure-based pump, flow sensors, microfluidic chips, reservoirs, and all other accessories required to operate within a cohesive software environment. The pack allows a researcher to move past the unboxing phase and begin active experimentation with minimal setup time.
What microfluidic chip is to be used in the process of liposome synthesis, and why?
The herringbone micromixer, which has two inlets and creates chaotic advection even at the lowest Reynolds number (below 1), is the standard chip being used. Such a turbulent mixing state ensures rapid and homogeneous blending of the lipid-ethanol phase with the aqueous phase, thereby initiating nanoprecipitation and LNP self-assembly. This approach offers better encapsulation efficiency and smaller size control compared to bulk methods. In low-flow-rate applications, co-flowing or flow-focusing chips may be used to maximize production yield.
What are the common uses of microfluidic LNP production?
LNPs prepared using this pack are applicable in a wide range of biomedical and cosmetic sector such as:
- siRNA and RNAi delivery: LNPs carry small interfering RNA to suppress gene expression that contributes to disease.
- Development of mRNA vaccines, such as BNT162b2 (BioNTech/Pfizer) and mRNA1273 (Moderna), both use LNPs to deliver mRNA to host cells.
- Treatment of polyneuropathy and antitumor, with LNPs as drug carriers,
- Solid lipid nanoparticles for cosmetic active ingredient delivery.
Why should the pressure-driven flow control be used instead of syringes or peristaltic pumps?
The nature of pressure-driven systems is inherently that of a pulseless supply of material to the microfluidic channel, i.e. providing a continuous flow that is perfectly smooth. This is very essential in liposome production since any slight variation in the flow will lead to changes in the ratios of mixing as it influences the particle size and polydispersity. Furthermore, push controllers will reach the desired flow rate much quicker than the syringe or peristaltic pumps, minimizing the transient effects at the beginning of every experiment. The flow controller and inline flow sensor can be used together to establish a real-time feedback loop, which allows the most accurate flow regulation possible in the market.
What is the size of the particles that can be produced using this system?
Liposomes and LNPs with mean diameter ranging between 20 nm and 100 nm have successfully been synthesized using the same configurations as these in this pack. Pharmaceutical uses of this range are of special interest, with particles within this range typically being ideal in systemic drug delivery, cellular absorption and evading renal clearance. Tuning the size is possible by modifying the flow rates and the flow rate ratio (FRR) of the organic and aqueous phases.
Does the system allow a scale-up in the laboratory to larger volumes?
The pack is constructed with a simple scaleup as one of its foundations. Increasing throughput can be achieved by:
- Increasing volumetric flow rates within known operating limits,
- The capacity to parallelize several herringbone micromixers at the same time using Tjunctions or 9manifolds that multiply the volume of output but keep the same mixing conditions in every single chip.
The strategy maintains monodispersity and encapsulation efficiency, and it can be applied to the production of liposomes at volumes ranging from microliters to liters.
What is the biological cargo that can be incorporated into the LNPs?
Hydrophilic molecules to be encapsulated in the formed LNP can be introduced into the aqueous phase of the herringbone chip. Examples recorded are:
- siRNA, which is encapsulated when the lipid shell spontaneously assembles around the aqueous core throughout nanoprecipitation,
- mRNA, as used in vaccine formulations,
- Other therapeutic payloads (nucleic acid constructs or small molecules) dissolved in the aqueous phase.
A higher encapsulation efficiency than the bulk approach is typically achieved with the microfluidic approach, due to more controlled and faster mixing dynamics.
Is the pack adaptable to special experimental requirements?
Yes. The pack is designed in a strictly modular, adaptable way. MIC professionals collaborate with every client to set up the following:
- Number of pumping channels (depending on the number of fluid streams that the experiment needs),
- Type of flow sensor (corresponding to the flow rate range of the use),
- Chip geometry (herringbone, flowfocusing, or coflow, depending on throughput and target particle size),
- Unless provided otherwise, chemical reagents (lipids, surfactants, buffers).
The team also includes elaborate synthesis protocols as part of the setup assistance. It is still in the beta testing stage, which implies that users enjoy intimate partnering with MIC during implementation.
What is the role of MIC as a member of Horizon Europe consortia?
In a Horizon Europe project consortium, the Microfluidics Innovation Center (MIC) positions itself as a special SME partner:
- A non-academic beneficiary bringing cutting-edge microfluidic instrumentation and technical expertise to research networks,
- A provider of demonstration and validation platform, especially in nanoparticle synthesis, organ-on-chip, or biosensor integration activities in the project,
- An actor in technology transfer, assisting in transforming academic outcomes into experimental platforms that can be deployed.
In applications that involve microfluidic expertise, such as LNP production in nanomedicine, on-chip cell culture, or diagnostics lab-on-chip, MIC has the equipment and application experience needed to enhance the technical standing and implementation strengths of a consortium. MIC can be approached by researchers and consortium coordinators to discuss the form a collaboration might take within a particular Horizon Europe call.
What support services does MIC offer?
Since the pack is in a beta testing program, early adopters receive support through MIC, which includes:
- Ondemand access to application scientists to develop protocols as well as for troubleshooting,
- Recommendation on how to maximize flow rate ratios, choice of chip, and preparation of reagents.
- The Galileo user interface is a softwarebased tool that centralizes control of all instruments in the pack.
- Help with the design of experiments for researchers unfamiliar with microfluidic LNP production.
Interested users can contact our team to find out whether they can join the beta program and receive an individual quote for their specific applications.

