Tips & Tricks for a successful HORIZON-HLTH-2026-01-DISEASE-04 proposal
Opening
10 February 2026
Deadline
Keywords
Cluster health
viral pathogens
epidemic potential
novel vaccine
clinical safety studies
health security
clinical translation
GMP readiness
immune profiling
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
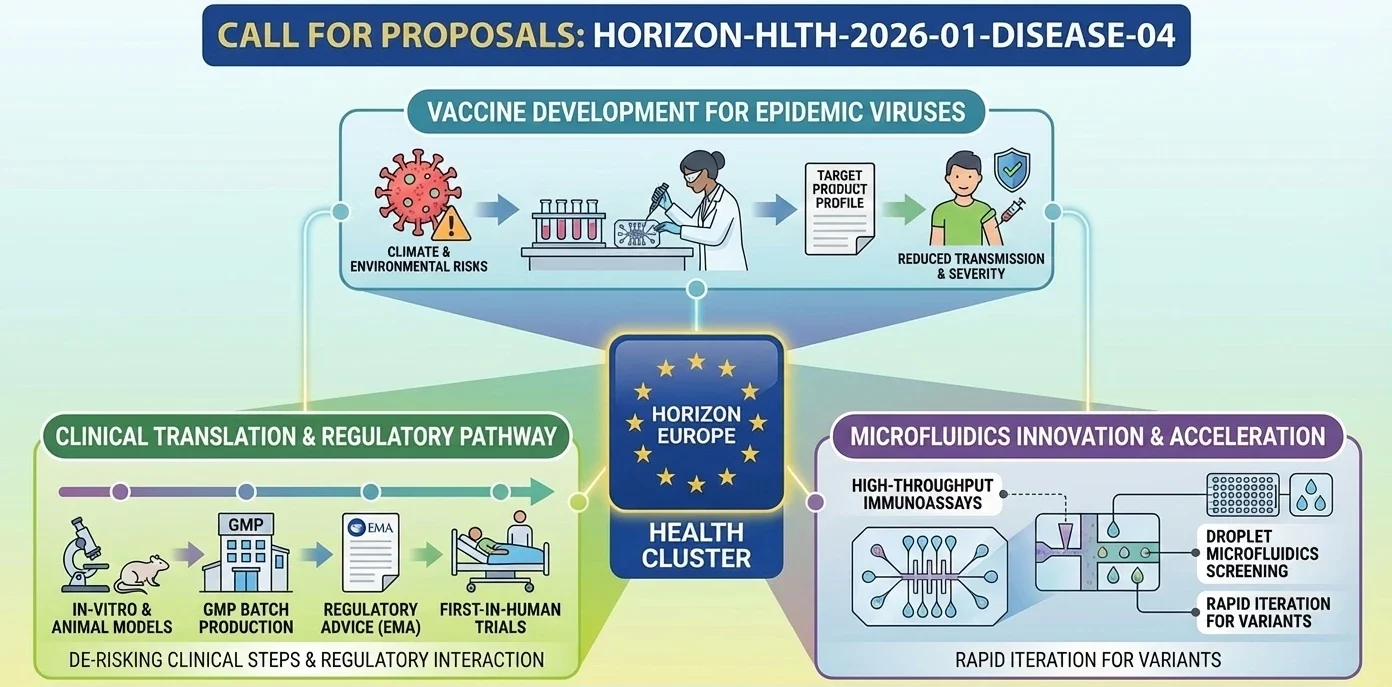
HORIZON-HLTH-2026-01-DISEASE-04: Development of novel vaccines for viral pathogens with epidemic potential
The European Commission has the desire to have new vaccine candidates against the viruses that may cause the next epidemic. Viral emergence is increasing quicker than ever due to climate change, with the solution in the EU being to invest in early-stage vaccine research today before an outbreak compels the agenda. This subject lies within the bigger European Medical Countermeasures Strategy. The objective: make experimental vaccines progress down the pipeline to the point that they can be used quickly in case things go awry.
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2026-01-DISEASE-04 call?
Which call is it, and when is the opening and the deadline?
- Call: HORIZON-HLTH-2026-01, Cluster 1 Health (Single stage 2026)
- Destination: Tackling diseases and reducing disease burden
- Topic: HORIZON-HLTH-2026-01-DISEASE-04
- Opening date: 10 February 2026
- Deadline: 16 April 2026 (17:00:00 Brussels time)
- Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
- Overall topic budget: EUR 44.20 million
- Number of projects expected to be funded: 5
- Expected EU contribution per project: EUR 9.00 to 11.00 million (roughly EUR 8.84 million per project if divided equally)
What are the key eligibility and evaluation conditions?
- Thresholds: 4 (Excellence), 4 (Impact), 4 (Implementation). Cumulative threshold: 12.
- Portfolio balancing applies: grants will be awarded not only by overall ranking but also to the highest-ranked proposal per targeted virus, as long as it passes all thresholds.
- US legal entities are eligible for EU funding.
- Legal entities established in China are not eligible.
- The JRC may participate as a beneficiary with zero funding or as an associated partner (not involved in proposal preparation or submission).
- Granting authority may object to IP transfer or exclusive licensing up to 4 years post-action.
- Subject to restrictions for the protection of European communication networks.
Deadlines of European Programmes 2026/2027
Get the MIC Horizon Europe 2026/2027 Calls Calendar:
-All Horizon Europe deadlines (by cluster and call).

Scientific range: what does the Commission expect from the HORIZON-HLTH-2026-01-DISEASE-04 grant?
It is a specific, targeted call to pathogens. Each proposal has to be specific to a single virus, in a list of specified viruses. None of the multi-target platforms attempting to do it all simultaneously. The single-virus depth, rather than breadth, is after which the Commission is pursuing.
Eight of the qualified viruses include Junin mammarenavirus, Lassa mammarenavirus, Andes virus, Hantaan virus, Sin Nombre virus, Hendra virus, Enterovirus D68, Venezuelan equine encephalite virus.
The work programme anticipates propositions to cover all the following:
- In-vitro characterisation of vaccine candidates that are under development (target specificity, epitope, ability to block viral action)
- In-vivo experiments with at least one animal model, or humanised immune system models, with sufficient protection to warrant direct transfer to humans.
- Non-human primate The regulators may need them before clinical trials, so the primate studies.
- The most promising candidates should be produced in large batches by GMP.
- First-in-human clinical safety trial with an apparent regulatory route in market approval.
To top all that, the Commission would like the biological sex, age, ethnicity, disability and social determinants which influence immune response and vaccine uptake to be addressed. This is not by the way in the anticipated results. Reviewers will look for it.
The input of countries where the targeted virus is endemic is welcome. And projects will take advantage of the existing research infrastructures and especially those related to the ISIDORe network. The first interaction with regulatory agencies does not happen by chance.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-HLTH-2026-01-DISEASE-04?
What are the most important scientific options?
- Choose your virus wisely. The portfolio-balancing rule implies that there is competition among the viruses, not only in the entire budget. A good proposal on a less popular virus (Hendra, say, or Venezuelan equine encephalitis) can be more successful than the fifth-best proposal on Lassa.
- Demonstrate a positive regulatory direction. This is not basic discovery that is being funded by the Commission. They desire applicants which already possess a certain preclinical foundation and have the capability of approaching first-in-human safety clinical trials within the lifetime of the project. We have observed appraisers punishing applications in which the regulatory plan appears to be open-ended or dreamy.
- Gmp production is not an option but a requirement. Budget it properly. The reviewers understand the cost of GMP batches and the fact that you are undercosting your manufacturing plan is an indicator that you do not fully understand the pipeline.
- Do not consider the sex and social determinants angle as a backdrop. The anticipated outcome text refers to the knowledge on the effect of biological sex and social factors on immune response, efficacy, safety, uptake. This is to be included in your study design, not in a WP9 paragraph on dissemination (check twice).
- Specifically use the ISIDORe infrastructure connection. Name the platforms. Discuss their integration into your experiment. No one will get impressed by it referring to the European infrastructures in a generic manner.
- Another aspect: the JRC has shown an interest in in-vitro biotechnologies on this issue. In case there is a natural fit with your approach, you can include the involvement of JRC in your consortium and this may enhance your proposal.
Consortium & proposal-writing plan: what works best with this type of Health RIA?
- In the case of EUR 9 to 11 million over most likely five years, we would suggest about 8 to 12 partners. Suffice to take virology, immunology, animal modeling, GMP production, clinical trials management, regulatory. Not too many that organization consumes your funds.
- One of your partners must have a BSL-3 or BSL-4 capacity, depending on whatever virus you have. And at least one clinical location with experience conducting Phase I trials in the target population of the infectious disease vaccines.
- Provided that your virus is endemic in a non-EU country then bring a partner in that country. The work programme states that it is encouraged and this phrase in EU grant language usually implies that you will score higher in case you do.
- A new SME that has technology of a vaccine platform or GMP ready production facility is a big burden. Evaluators appreciate the fact that the project will be able to generate something at the end not merely publish results. SMEs also are a pointer of commercial follow-through.
- Write the section of impact about epidemic preparedness, HERA, and Medical Countermeasures Strategy. Bury these associations not in a table. Put them in the narrative.
- To the implementation section, your Gantt chart must demonstrate clear regulatory milestones. At every stage, evaluators will seek a go/no-go logic. When you are thinking of doing preclinical, then clinical, then publish, that is too linear, and likely overly optimistic.
How would microfluidics contribute to this topic?
The development of vaccines to deal with rare or emergent pathogens hits a practical limit at an early stage: you cannot always have enough samples of patients, you cannot always establish studies in large animals in a short period of time and you can sometimes not know about immune responses that cohort data can not tell you. Microfluidics occupies some of those gaps in a manner making it difficult to substitute them.
- Chip based immune response profiling. Suppose you are interested in understanding the response of a candidate vaccine in T-cell or B-cell of donor backgrounds. You can perform those assays on an organ-on-chip platform with very small sample volumes which is important when you are dealing with pathogens where clinical material is limited.
- Accelerating the screening of antigens. Droplet microfluidics are able to produce and screen thousands of antigen variants at once. In a virus such as Hendra or Venezuelan equine encephalitis where the pool of candidates remains small then this type of throughput can provide you with an actual lead in the right construct.
- Microfluidic tools can be used to model the mucosal barriers or vascular tissue exposed to the virus to determine whether the immune response induced by the vaccine really protects the tissue of interest. Same compound, different tissue situation, different solution.
- Formulations development that is GMP-compatible. Lipid nanoparticles and other delivery vehicles are reproducibly produced in microfluidic mixers of batch after batch. You get one answer twice and this is what regulators would want to see.
- On the sex-specific immunology front microfluidic systems can take male and female donor cells with the same conditions and allows you clean comparisons which animal models and makes untidy.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ - HORIZON-HLTH-2026-01-DISEASE-04
What are the appraisal levels, and how does it work in real life?
Each of the criteria (Excellence, Impact, Implementation) must be scored at least 4/5, and the overall score must be 12/15. In practice, a single poor evaluation requirement can doom a good proposal, so the scientist (Excellence), the health-security policymaker/market shaper (Impact), and the project manager/regulator (Implementation) must all be represented well.
Does it fund based on rank or portfolio rule?
Explicit portfolio logic: to cover target viruses, the Commission can fund the best proposals in each area of selection, as long as there is a threshold. Translation: it is as important to be best-in-class for your preferred virus as it is to be ranked globally. Be clear about your decision and explanation of the virus.
What will be the scientific deliverables at the conclusion of the grant?
Protective and safe vaccine candidates that are developed to a degree where bigger clinical trials can be used. The work should include non-clinical packages of work with a coherent focus (e.g., in vitro characterization and pertinent animal models), GMP-scale-up production/batching of clinical material, where necessary, and first-in-human safety studies. The final product must be presentable as a regulator-ready product to the next step, rather than a prototype seeking a plan.
What should we do with our R&D plan to make it look fundable and interesting?
Ground the proposal on an evidence-based clinical translation path and an obvious Target Product Profile. Present dose rationale, stopping criteria, safety surveillance; match non-clinical trials to the target clinical population and clinical endpoint; present target decision-gate criteria that were measurable go/no-go. A pipeline diagram (1 page) that includes inputs, WPs, milestones, and readiness levels gives evaluators, at a glance, a sense of coherence.
In what ways can microfluidics be used to enhance a DISEASE-04 proposal?
Microfluidic systems are capable of condensing timeframes and minimizing sample sizes, such as in times of epidemics:
-On-chip neutralization and immune-profiling ranking of antigen/adjuvant options at higher throughput rates.
-Droplet microfluidics for rapid formulation screening and antibody response mapping.
-Automation-friendly, standardized, and reproducible workflows of analysis that enhance reproducibility across locations, as well as rapid data lock.
To put it into perspective, our team at the Microfluidics Innovation Center (MIC) designs end-to-end microfluidic systems, produces custom chips, and constructs prototype instruments on demand; these resources typically enhance the value of Excellence (vigorous science) and Implementation (rapidity of execution). Multi-partner consortia usually serve as a broker between immunology laboratories, GMP manufacturers, and clinical communities.
What consortium structure will score this RIA highly?
-Plausible chain of command between the bench and the bedside:
-Antigen design, immunology, assay development, and thinking about correlates of protection: vaccine R&D.
-Preclinical: access to proper animal models and challenge facilities; sound biostatistics.
-Manufacturing: collaborates with GMP preparation, process development, and scale-up reasoning (direct or subcontracted).
-Clinical: a group that is capable of conducting real first-in-human safety trials, taking into account such aspects as ethics, pharmacovigilance, and trial operations.
-Regulatory: A person who has already passed through EMA/national advice and initial trials.
An innovative SME: introduces speed, product attitude, and specificity of exploitation. Intuition suggests that introducing a specialized SME, such as MIC, into a Horizon Europe health consortium is much more efficient and, according to our internal monitoring, the odds of success are about twice as high as for comparable calls, as described in the official baseline reports.
Any writing strategies that can prompt evaluators to score quickly and generously?
Yes, reflect the Excellence/Impact/Implementation structure and create all subsections that can be scored. -Introduce reader-friendly mechanisms: a one-page graphical representation of the development pathway; clear and numeric go/no-go criteria (e.g., neutralization titer fold-rise laws, manufacturing productive capacity in each batch, equitable access); and an Impact section aimed at outbreak decision-makers (deployment turnaround, manufacturing responsiveness, fair access), rather than just academic colleagues.
What of TRLs, IP, data, and ethics is enough?
-Define your initial TRL, your desired final project TRL, and the change in the needle by each of your work packages.
-IP foreground/background and access rights map early to eliminate ambiguities during exploitation.
-Pledge to FAIR principles and safe management of clinical and genomic data, and a realistic data-management plan for data. The ethics areas are expected to address first-in-human safeguards, the inclusion of underserved populations for reasonable reasons, and dual-use/biosecurity.
What is the way to capitalize on the portfolio rule instead of being afraid of it?
Select an objective virus where your team is arguably best. Then, facilitate easy selection of the Commission to ensure equal distribution: demonstrate spotless milestones, schedule actual GMP and first-in-human studies, and outline a backup plan if an early candidate does not perform. And you would like people reviewing to think: “This is the surest bet in this area of viruses; they will come.”
What is the role of MIC with a DISEASE-04 project?
MIC excels in microfluidic and automation, providing design and manufacturing of custom chips, high-throughput immunoassays, droplet screening systems, instrument prototypes, and powerful analytical SOPs for partners. MIC entry success in recent consortia has nearly doubled the odds of success, despite the comparison of baseline acceptance rate and simple manufacturability and end-user validation rates, as these are accommodated during the very first month of consortium initiation and never tacked on in month 24.
Last-minute checklist- what should not be left out?
-The selection of the virus and rationale is evidently linked to EU preparedness.
-TPP has quantitative endpoints (immunogenicity, dose range, concentration range, manufacturing yield).
-GMP plan/ batch release testing strategy.
-First-in-human design synopsis, risk mitigation, and DSMB plan.
-Pre-regulatory-advice schedule and questionnaire.
-Go/no-go gates are numbered (Milestones).
-Exploitation route: scale-up, manufacturing partners, and route to pivotal trials.
-A one-page pipeline chart that any reviewer can read within 30 seconds.
