Tips & Tricks for a successful HORIZON-HLTH-2027-03-TOOL-04 proposal
Opening
03 June 2027
Deadline
Keywords
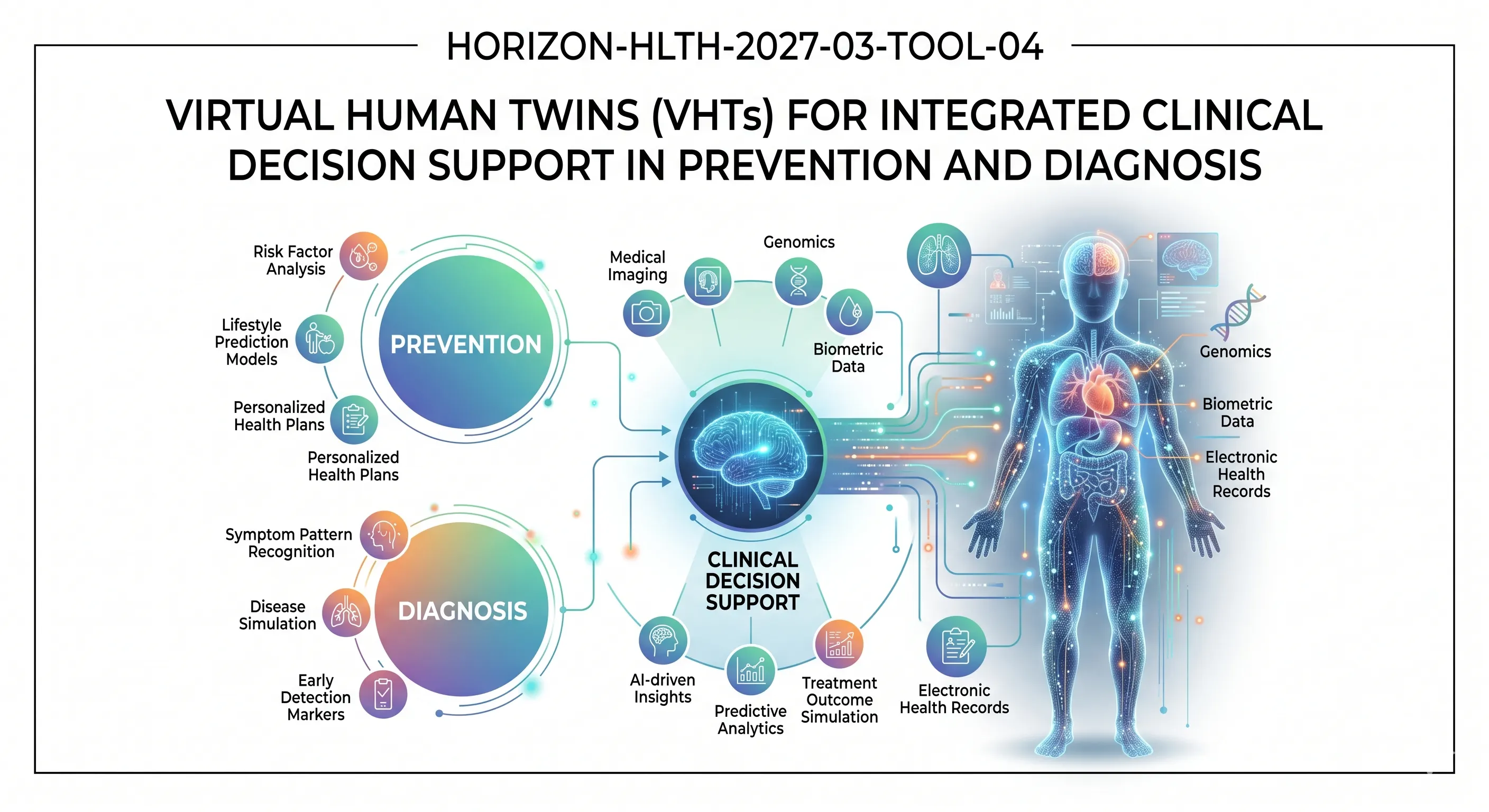
Virtual Human Twin
Multi-organ modelling
genomics
Clinical decision support
Personalised medicine
In-silico modelling
Digital health AI
disease simulation
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
HORIZON-HLTH-2027-03-TOOL-04: Virtual Human Twins (VHTs) for integrated clinical decision support in prevention and diagnosis
The Commission would desire Virtual Human Twin (VHT) technology to move out of the laboratory to the real clinical care settings. This is not a request to provide more proof-of-concept models, but rather to roll out some genuinely working, patient-specific, multi-organ computational systems that can aid physicians in making decisions in the prevention and diagnosis of high-burden disease domains. This necessitates strong clinical evidence, health economic evidence, and proven incorporation in care pathways.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2027-03-TOOL-04 call?
Which call is it, and when is the opening and the deadline?
- Call name: Cluster 1 – Health (Single stage – 2027/2)
- Call identifier: HORIZON-HLTH-2027-03
- Destination: Developing and using new tools, technologies and digital solutions for a healthy society
- Topic: HORIZON-HLTH-2027-03-TOOL-04
- Opening date: 03 June 2027
- Deadline: 22 September 2027 (17:00 Brussels local time)
- Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
- Total indicative topic budget: EUR 39.30 million
- Expected EU contribution per project: EUR 10.00 to 12.00 million
- Indicative number of projects funded: 4
What are the key eligibility and evaluation conditions?
- Award thresholds: 4/4/4 (Excellence, Impact, Implementation); cumulative threshold of 12
- Special eligibility: legal entities established in the United States of America are eligible to receive EU funding, in recognition of NIH reciprocity
- If satellite data are used, beneficiaries must use Copernicus and/or Galileo/EGNOS
- Subject to restrictions for the protection of European communication networks
- SSH (social sciences and humanities) expertise required in the consortium
- Clinical studies annex must be provided using the submission system template
- Budget must be reserved for contributions to the Platform for Advanced VHT Models and the European VHT Initiative
- SME participation explicitly encouraged; no JRC involvement flagged for this topic
Scientific range: what does the Commission expect from the HORIZON-HLTH-2027-03-TOOL-04 grant?
What outcomes are expected?
By the project completion, clinicians should be equipped with validated models of multiple organs that are patient-specific and which can assist in the active decision-making in prevention and diagnosis within a healthcare environment, and not within a laboratory. The Commission is seeking health economic evidence of cost-effectiveness, and a clear roadmap on regulatory compliance. These patient-specific prevention and diagnostics tools are to be implemented in patients with different features, such as sex, age, and ethnicity.
What is within scope?
Multi-scale (between sub-cellular and organ systems), multi-organ, and longitudinal VHT of any high-burden disease area is covered by the call. Specifically, the work programme discusses: comorbidities, chronic cardiovascular conditions, infection and autoimmunity, inflammation and cancer, diabetes, rare diseases, degenerative diseases and exposome-related health impacts. The computational approaches can be biophysics-based modeling, interpretable or explainable AI, generative AI, in-silico modeling, agent-based approaches, and network physiology. It does not focus on pure disease management (as in 2023 predecessor topic).
What are the specifically proposed research directions?
- Clinical use cases based on the multi-organ and multi-scale time-dependent modeling of the complex disease processes are to be validated and selected.
- Creating and designing dynamic computational models that are based on existing data resources (biobanks, EDITH CSA outputs, Platform for Advanced VHT Models).
- Conducting cross-modeling across the various modeling methodologies, integrating and validating them into interoperable, patient-specific VHT models, i.e. showing that VHT models can be used with current preventive and diagnostic tools across the care pathways.
- Creating clinical validation data and health economic data (cost-effectiveness in comparison with current practices).
- Development of an exploitation plan, which deals with regulatory compliance, and intellectual property.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-HLTH-2027-03-TOOL-04?
What scientific choices matter most?
- Choose a practical clinical case with significant morbidity burden: The characteristics of high need and multi-organ, multi-scale modeling will be sought in disease areas that have high and unmet needs and where such modeling is indeed required e.g. complex comorbidities like diabetes combined with cardiovascular disease, or neurodegenerative diseases in interaction with mental health.
- Don't just build the model; demonstrate its clinical utility: From the beginning, physicians should be engaged. Co-design the work plan with clinicians, not only as a post-hoc dissemination step.
- Ensure AI components are explainable: The labor plan is self-evident; AI has to be readable or understandable. It will not accept black-box prediction engines. This is to be incorporated in your technical design, integrated with the European VHT ecosystem or not.
- Connect to the European VHT ecosystem or don't: The projects must be consistent with the EDITH CSA, the Platform of Advanced VHT Models, the 1+Million Genomes Initiative, and EU4Health projects. Otherwise, you will give an impression that you are not doing your homework.
- Collect robust health economic data: Cost-effectiveness analysis is critical in the real world. This is what should be included in your assessment plan and you must find clinical partners to help in this.
- Proactively include sex, age, and ethnic diversity: These aspects must be factored in the model design, and not thought of as an afterthought during the discourse. This is one of the pitfalls of most consortia.
Consortium & proposal-writing plan: what works best with this type of call?
- Target a consortium of 10-14 partners, potentially slightly more if clinical coverage demands it.
- Include computational modelers, clinicians with patient data access, at least one health economics team, and SSH researchers. These roles need not be necessarily big university roles. Preferably, engage two or three clinical sites of Member States.
- Ideally, involve at least two or three Member States’ clinical sites. This reinforces the argument on generalizability by proving that it has a diverse patient population and addresses the diversity requirements.
- Add a new SME, preferably with software development or medical equipment experience. The work programme literally promotes the participation of SMEs, and a realistic commercial exploitation route will be positively welcomed by the evaluators.
- If you have US-based data or NIH collaborators, consider the US eligibility exception. This has the potential to be very beneficial to certain consortia.
- When writing your proposal, adopt the language and framing of VHT. The Commission has put a lot of money into the VHT concept and has invested it on multiple cycles. Quote EDITH, see the Platform of Advanced VHT Models, and identify with the European VHT Initiative. Not doing so will give an impression that you have not done your research.
- Prepare the clinical studies annex early. People do not realize that it is more time-consuming.
- Frame your impact section around deployment barriers rather than scientific novelty. How your project will not end up in a dustbin (stagnating in the drawers) is more valued by evaluators than the complexity of your model.
How would microfluidics contribute to this topic?
Standard computational models of disease rely on population-level data and average tissue parameters. The problem is that individual patients don't behave like averages. Microfluidic organ-on-chip systems generate patient-specific, dynamic biological data at the tissue and cellular level that conventional in vitro models can't provide. That data is exactly what VHT models need to become genuinely patient-specific rather than just demographically adjusted.
- For example, in building a VHT for a patient with cardiovascular disease coupled with a chronic inflammatory disease; in order to obtain blood vessel wall responses to that specific patient's immune system, that patient's cells will be used to seed a vascular chip, and an experiment performed, yielding calibrating data which cannot be generated in any other way in silico.
- Multi-organ coupling requires interface data: gut-liver and kidney-cardiovascular interactions cannot be reasonably simulated computationally, without measurements of transference parameters, which can be obtained in an organ-specific, controlled, and reproducible manner through multi-organ microfluidics, under variable experimental conditions.
- Microfluidics could also play an important role in validation, so that prior to human trials, your consortium will have tested if the VHT's predictions regarding drug response or disease development can be supported by observing the response of actual tissue under physiological challenge; results will be repeatable.
- With regard to use case in rare diseases: small patient populations and lack of longitudinal data can lead to difficulties with model training and validation with clinical data, something a chip model can potentially address where VHT literature largely ignores this; this gap would also be significant for any potential consortium.
Microfluidics is not a substitute for the computational core of a VHT but would make for a stronger proposal by bridging an experimental (chip) and computational (model) system, using data rather than estimates. MIC could act as a bridge between the chip and the model in a consortium geared toward clinical validation.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ – HORIZON-HLTH-2027-03-TOOL-04
What type of action is HORIZON-HLTH-2027-03-TOOL-04 and when is the deadline?
This is a Research and Innovation Action (RIA) of Horizon Europe Health Cluster 1, call HORIZON-HLTH-2027-03. It will start on 03 June 2027 and finish on 22 September 2027 at 17:00 local time in Brussels.
What is the budget per project for this VHT call?
The indicative budget of the topic is EUR 39.30 million, and it is anticipated that the project will receive between EUR 10.00 and 12.00 million as the EU contribution. About four projects will be financed.
What exactly is a Virtual Human Twin (VHT) in this context?
A Virtual Human Twin (VHT) is a computer-based, in-silico model of the health and illness of a particular patient, at various levels of human anatomy, such as sub-cellular through to organ-system levels. This call is specifically focused on applying these models to aid in prevention and diagnostic decision-making on an even more complex level than the 2023 predecessor subject.
Which disease areas and clinical use cases are in scope?
The Commission is looking for multi-scale, multi-organ, longitudinal approaches. Clear areas of disease are comorbidities, chronic cardiovascular disease, infection and autoimmunity, inflammation and cancer, diabetes, rare diseases, degenerative diseases, and exposome-related health effects.
Is SSH expertise really required in the consortium?
Yes. Explicitly required in the consortium is SSH (social sciences and humanities) expertise. Proposals will also be user-friendly (end-user) and will incorporate clinician and patient feedback throughout the design and development process.
Can US-based institutions participate in this topic?
What EU initiatives must the project connect to?
It is anticipated that projects will align with the Platform for Advanced VHT Models, the European VHT Initiative, the EDITH CSA, the 1+Million Genomes Initiative, and other activities of the EU4Health and Digital Europe Programs. These contributions need to have a separate budget line.
Why is SME participation so important for this call?
The involvement of SMEs is clearly promoted. Creative SME in software development or with experience in a medical device enhances the technical credibility as well as the commercial exploitation plan- two aspects that the evaluators will be keen on when an RIA of this magnitude is being reviewed.
How can microfluidics contribute to Virtual Human Twin development?
Microfluidic organ-on-chip systems generate dynamic, patient-specific biological data at the cellular and tissue levels. This information corrects and legitimizes VHT models and they are truly patient-specific. Multi-organ microfluidic systems also generate the interface-transfer parameters required to model organ crosstalk, which computational methods cannot reliably provide.
How can MIC support a HORIZON-HLTH-2027-03-TOOL-04 proposal?
MIC has been involved in several Horizon Europe proposals for organ-on-chip and multi-organ systems. MIC, as an innovative SME, will provide implementation capability, proven microfluidic platforms, and proposal-writing experience to any consortium seeking to apply to HORIZON-HLTH-2027-03-TOOL-04.
