Tips & Tricks for a successful HORIZON-HLTH-2027-02-TOOL-01-two-stage proposal
Opening
10 February 2027
Deadline
Keywords
omic data
clinical use
Personalised medicine
AI methods
treatment response
disease progression
Multimodal biomarkers
Chronic NCDs
Real-World Data
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
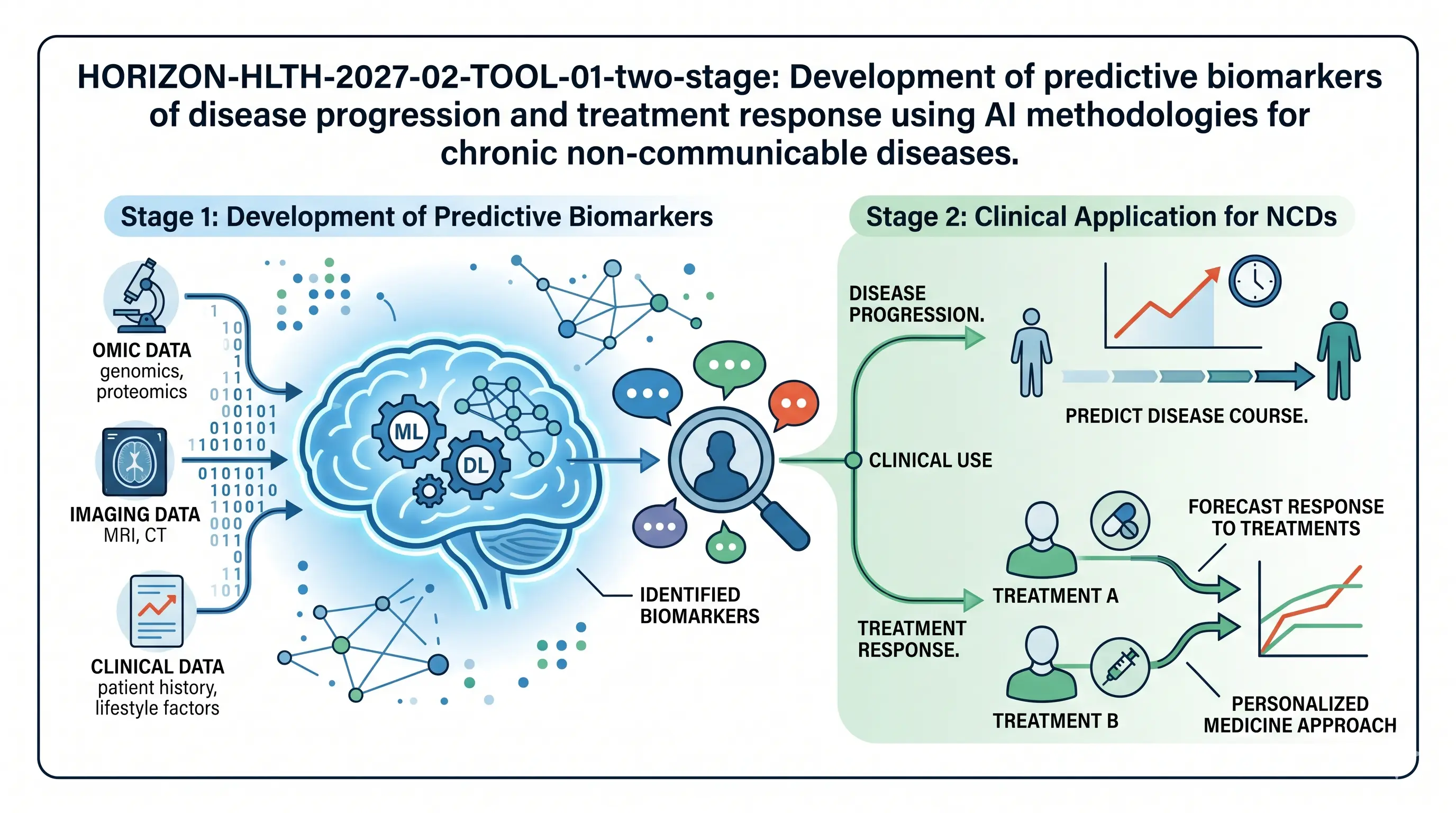
HORIZON-HLTH-2027-02-TOOL-01-two-stage: Development of predictive biomarkers of disease progression and treatment response by using AI methodologies for chronic non-communicable diseases
One more of the gaps that the Commission would like to seal is that over the years there has been a discovery of thousands of biomarkers, and only a handful of them ever made it to clinical use. The concern of this call is to apply AI to fuse, prioritise and validate predictive biomarkers of non-communicable diseases to enable the clinician to have an improved way of tracking the progression of a disease and whether a treatment is working in the future. Cancer is evidently shunned.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2027-02-TOOL-01-two-stage call?
Which call is it, and when is the opening and the deadline?
- Call name: Cluster 1 – Health (Two stage – 2027)
- Call identifier: HORIZON-HLTH-2027-02-two-stage
- Destination: Developing and using new tools, technologies and digital solutions for a healthy society
- Opening date: 10 February 2027
- First stage deadline: 13 April 2027
- Second stage deadline: 22 September 2027
- All deadlines at 17:00 Brussels local time
- Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
- Overall topic budget: EUR 44.20 million
- Indicative number of projects to be funded: 6
- Expected EU contribution per project: EUR 6.00 to 8.00 million
What are the key eligibility and evaluation conditions?
- First stage is evaluated blindly. Applicants must not disclose organisation names, acronyms, logos or personnel names in the abstract and Part B of the first-stage application.
- First stage thresholds: 4 (Excellence) and 4 (Impact). The overall threshold is set so the total requested budget admitted to stage 2 is as close as possible to four times the available budget, and not less than three and a half times.
- Second stage thresholds: 4 (Excellence), 4 (Impact), 4 (Implementation). Cumulative threshold: 12.
- US entities are eligible for EU funding (NIH reciprocity clause).
- Restrictions for the protection of European communication networks apply.
- Granting authority may object to transfer of ownership or exclusive licensing of results up to 4 years after the end of the action.
- SME participation is encouraged. Clinical studies template is strongly encouraged.
- Funded projects will be strongly encouraged to participate in networking and joint activities with other selected projects. Budget should be allocated accordingly.
Scientific range: what does the Commission expect from the HORIZON-HLTH-2027-02-TOOL-01-two-stage grant?
What outcomes are expected?
The Commission is seeking a metamorphosis in the application of biomarkers in non-communicable diseases. The clinicians should be in a position to have access to validated predictive biomarkers which can present something about disease progression and response to treatment as opposed to establishing a diagnosis at the end of the project. Artificial intelligence machines should be useful, robust and free clinical trials should be carried out. The reason behind this is that the ultimate goal is to achieve better patient outcomes in personalised medicine whereby the clinical guidelines would be updated as to what the biomarkers can do.
What is within scope?
- Only chronic non-communicable illnesses. Cancer is explicitly excluded.
- Multimodal biomarkers that include molecular, cellular, physiological, imaging, behavioural and digital biomarkers as well as combinations of these.
- Use of developed AI techniques. The point that the Commission is clarifying here is that there is no longer a need to develop new architectures, but instead modify and deploy the existing AI. This is not what they are investing in.
- Combination of the existing biomarker data, the potential biomarker data of the previous clinical trials, longitudinal data and Real-World Data (RWD).
- Biological sex inequality. It is possible that gender-specific variables and sociodemographic factors may impact disease progression or treatment compliance.
- Clinical validation in independent cohorts and RWD. Future clinical trials should be headed by EU/EFTA or other Associated Country organizations.
- Biomarker qualification and alignment of AI tool regulation strategy and exploitation scheme on bioethics.
- GDPR compliance. The European research and data infrastructures under the Digital Europe Programme are to be used.
What are the specifically proposed research directions?
- Evaluate and strategize the groundwork of the current biomarkers in clinical practice and prospective biomarkers of the accomplished and ongoing studies. An organized inventory is a point of departure that the Commission would like to have.
- In the data types, adapt created AI to pool and prioritize biomarkers. It is concerned with credibility: the measurement of performance, reliance, repeatability of the identical response, and detection of bias in the datasets.
- Implement a system of biomarker validation. This is likely to be the most weighty deliverable. To prove these biomarkers work better than the existing ones, clinical validation studies in their independent disease cohort using RWD and exploratory trials.
- Install decision-support and visualisation tools that can in fact be used by clinicians to determine the plausibility of biomarkers within a population of patients. The Commission states user-friendly, fit-for-purpose, which in actual sense implies: do not provide clinicians with a black box.
- Prepare a roadmap and plan of exploitation. Engage regulators early. The exploitation in the EU should be prioritised. This is whereby most consortia fail to take seriously.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-HLTH-2027-02-TOOL-01-two-stage?
What scientific choices matter most?
- Select prudently your disease area: The call teaches that it is chronic non-communicable diseases (no cancer) which leaves you with cardiovascular, metabolic, respiratory, neurological, musculoskeletal, and some others. Your decision would have to show apparent unmet clinical need and above all availability of quality longitudinal data. The proposal will not have an AI component without the data.
- It is time to display your data: This is usually the situation with proposals: teams guarantee to generate a dataset during the project. This is quite clear in the work programme: the quality clinical data should be readily available, and the creation of the new data on a small scale to streamline AI. In case you are not able to prove that you can access data at the proposal stage, you will lose points.
- Market the AI but do not overmarket it: The Commission suggests adjusting and deploying the existing AI methods. Reviewers will frown upon proposals that will transform it into a computer science project. In this case, the AI is not an end. Put it as a way to an end.
- Make investments in the validation platform: There, one can find the impact story. It is demonstration that your predictive biomarkers are better than current clinical practice in an independent cohort that makes the difference between a good proposal and an average one.
- Sex is not a matter of choice when it comes to gender stratification: The work programme offers stratification by biological sex and hopefully, gender related variables. This is to be built into your study design, not as an afterthought.
- Credibility and prejudice evaluation of the AI tools: The set of Ethics Guidelines of Trustworthy AI is mentioned in the Commission. Your proposal should have a definite way of performance measures, bias and privacy adherence. Such is what appraisers look into twice (twice on your part, as well).
- Plausibility in clinical significance compared to statistical: The decision-support tools should have the capability to allow clinicians to determine whether a p-value represents a clinical sense of a biomarker prediction or not.
- Initial regulatory intervention: Do not put the regulatory roadmap on the exploitation chapter. Consult EMA or even national regulators at the first level. As we have seen, the proposals containing a letter of support by a regulatory body get better points.
Consortium and proposal-writing plan: what works best with this type of call?
- Concerning the actual number of partners, aim at between 8 and 12 or 13 at maximum in case your clinical validation design demands more. This is an RIA that has a maximum budget of EUR 8 million and hence you do not wish to overstretch it.
- You have to have clinicians who own the longitudinal cohorts. This is non-negotiable. No access to cohorts, no proposal. Find out which hospitals or registries have a long history of collecting information on your disease. They are the basis of the project.
- There should not be domination of AI and data science partners. A single or the two powerful groups. The Commission does not want a machine learning competition but rather clinical biomarkers that are demonstrated by AI.
- Add an innovative SME, preferably one that has prior experience in clinical decision-support software, biomarker assays development, or health data analytics. This puts the SME box that the work programme focuses on and gives it an actual exploitation path.
- In case you can invite a regulatory science partner or a liaison with EMA, do it. It makes the exploitation and sustainability sections heavy.
- This is a first stage that is blind. Part B will have to proceed single-handed on the scientific argument, and no institutional reputation to appeal to. The initial step and all sentences should be worth their existence. No insulation and no boilerplate.
- One of the credibility factors that they bring to your dissemination and clinical relevance narrative is patient organizations or patient advocacy groups. Don’t overlook them.
- Devote approximately 2% on networking and teamwork. The Commission strongly promotes it and looks forward to its inclusion in the work plan.
How would microfluidics contribute to this topic?
Most traditional biomarker discovery and validation uses are done on large volumes of samples, centralised laboratory equipment and batch processing. It is alright in a research hospital, but it renders it sluggish when you are trying to confirm multimodal biomarkers using different cohorts across countries. It is possible to change the logistics of this process with the help of microfluidic platforms.
- The case situation is that you are doing a validation study of a panel of inflammatory and metabolic markers in six clinical sites. Lab-on-chip system would enable each site to perform the same test on the same platform using a fraction of blood volume and yet the same answer will be obtained regardless of whether it is the first or second technician loading the chip. Such consistency is important when your AI is cross-site learning.
- Organ-on-chip models can actually tell whether a candidate biomarker is actually monitoring what is happening in living tissue. You expose a miniaturised scale of vascular tissue or lung epithelium to a drug, measure the response in the biomarker and establish whether such a response correlates with patient outcomes. It is a way of ensuring clinical plausibility, but not necessarily to the point of conducting an actual prospective trial.
- Considering the case of digital and physiological biomarkers, microfluidic biosensors in point-of-care sensors can capture real-time data streams that can directly feed into AI models. That would provide a way for your consortium to obtain longitudinal biomarker information outside the hospital, which is what the Commission is trying to achieve with Real-World Data integration.
- Assays on a single chip multiplexed. They can be run on the same device by your consortium rather than sending the samples to four different labs where they can be analyzed using four different markers. Reduced sample, reduced cost, reduced turnaround.
Microfluidics will not write your AIs, but it will provide your consortium with the computing infrastructure they need to generate stable, multimodal biomarker data of the size and quality this call demands. Such a standardisation in a proposal to engage six clinical validation sites in Europe is what will lead to the difference between a paper-based validation platform and an operational one. The MIC regularly joins Horizon Europe RIA consortia as the technical partner that delivers exactly this type of platform engineering.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ – HORIZON-HLTH-2027-02-TOOL-01-two-stage
What is the HORIZON-HLTH-2027-02-TOOL-01-two-stage call really about?
The HORIZON-HLTH-2027-02-TOOL-01-two-stage call supports the development and clinical proof-of-concept of predictive biomarkers for chronic non-communicable diseases, integrating multimodal data via AI. The Commission is seeking tools to predict how a disease will evolve and how effective a treatment will be, not just to diagnose the disease. Cancer is excluded from this topic.
Who can apply and how big are the projects?
Which diseases are in scope, and which are not?
In scope: cardiovascular, metabolic, respiratory, neurological, musculoskeletal diseases and other non-communicable chronic diseases. Excluded: cancer, acute and infectious diseases.
Why does the Commission insist on established AI methods rather than novel ones?
The Commission is supporting clinical biomarker validation, not AI. They want reliable AI methods that are well-adapted to the biomedical question, with performance metrics and bias tests. Proposals that make this a machine learning challenge will have their marks deducted for Excellence and Impact.
What does blind evaluation mean for the first stage?
In the first stage, applicants should not include the name or acronym of their home organization, their logo, or the names of people in their abstract or Part B. A strong science narrative is vital. No reputational shortcuts
How important is data access at the proposal stage?
The work program states that appropriate clinical data should be readily accessible at the time of the proposal; limited new data should be generated to optimize the AI. Without clear access to cohorts and Real-World Data, the project is not credible.
What kind of consortium fits this call best?
Target 8-12 partners, sometimes 13 if multiple clinical validation centers are involved. You must have clinicians with access to longitudinal cohorts, one or two top-notch AI groups, an innovative SME to lead exploitation, and, preferably, a regulatory science partner or EMA contact.
Why is sex and gender stratification non-negotiable here?
The work program specifically calls for stratification by biological sex and, if possible, the inclusion of gender and other sociodemographic factors. The reviewers will assess whether this is woven into the study design from the beginning rather than bolted on afterward.
How does microfluidics fit into a biomarker validation project?
Lab-on-chip technology based on microfluidics can provide reliable, multimodal biomarker measurements across multiple sites from small sample volumes. Organ-on-chips can be used to validate that biomarker candidates are representative of the tissue. Real-time longitudinal data can also be obtained in point-of-care devices with microfluidic biosensors.
What are the most common reasons proposals fail in this type of topic?
Lack of understanding of data access needs, excessive AI ambition detracting from clinical validation, poor-quality validation cohorts, a lack of sex- and gender-stratification, and a “plan B” approach to regulatory strategy. Limited early engagement with the EMA and lack of an exploitation partner conclude the list.
