Tips & Tricks for a successful HORIZON-HLTH-2027-01-DISEASE-05 proposal
Opening
10 February 2027
Deadline
Keywords
small molecule antivirals
epidemic preparedness
viral pathogens
antiviral therapeutics
disease burden
European Medical Countermeasures
clinical data
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
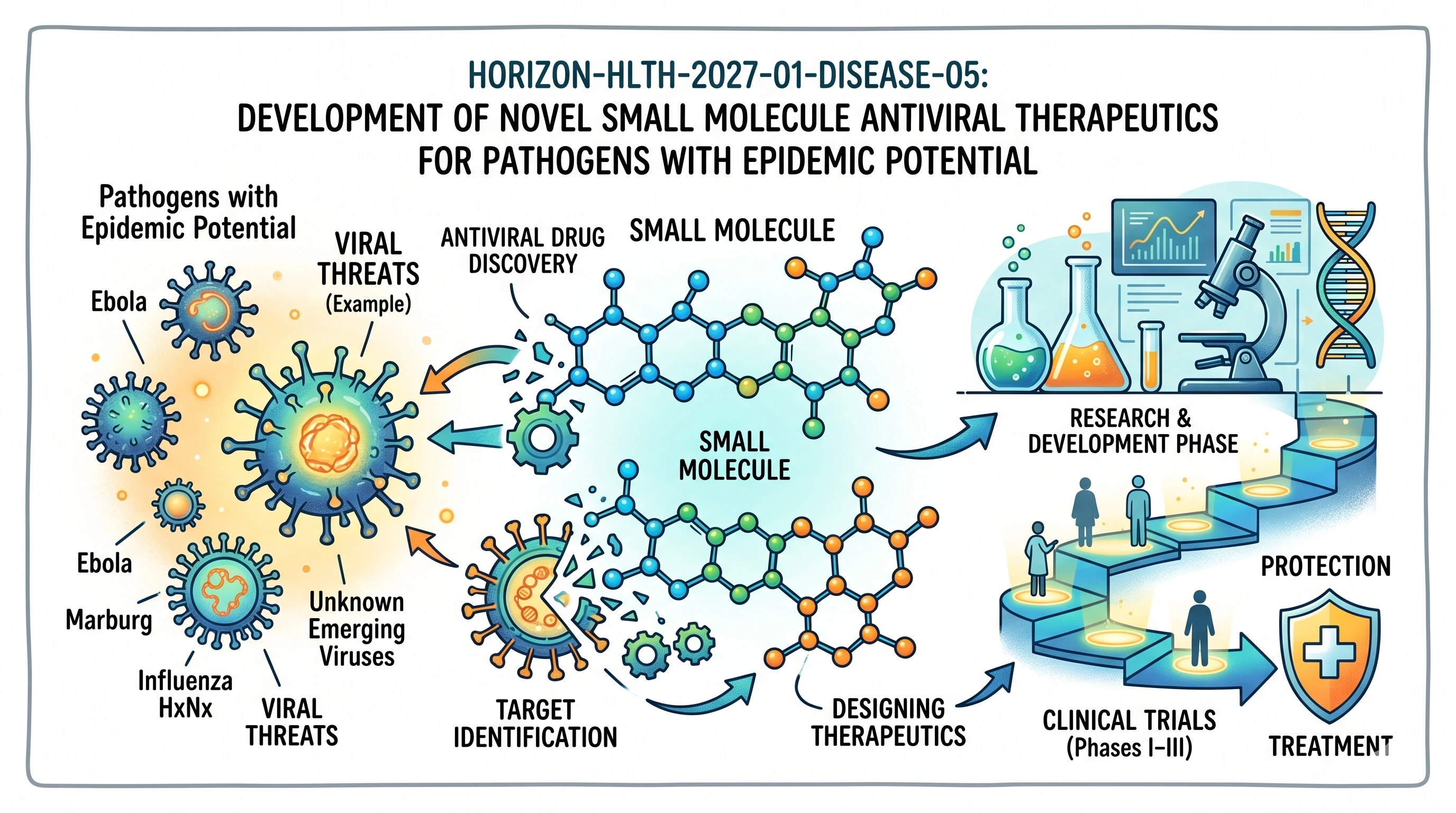
HORIZON-HLTH-2027-01-DISEASE-05: Development of novel small molecule antiviral therapeutics for pathogens with epidemic potential
This is not a blanket approach to antiviral research; the Commission is seeking to expedite already identified drug candidates to a shortlist of viruses deemed high-priority threats. This is part of the European Medical Countermeasures Strategy and is to achieve results that can be deployed in the event of an outbreak. By the end of the grant, clinical-stage data must be provided; promising lab data will not be accepted.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2027-01-DISEASE-05 call?
Which call is it, and when is the opening and the deadline?
- Call name: Call – Cluster 1 – Health (Single stage – 2027/1)
- Call identifier: HORIZON-HLTH-2027-01
- Destination: Tackling diseases and reducing disease burden
- Topic: HORIZON-HLTH-2027-01-DISEASE-05
- Opening date: 10 February 2027
- Deadline: 13 April 2027 at 17:00 Brussels time
- Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
- Overall topic budget: EUR 44.20 million
- Indicative number of projects funded: 5
- Expected EU contribution per project: EUR 9.00 to 11.00 million
What are the key eligibility and evaluation conditions?
- Thresholds: Excellence 4, Impact 4, Implementation 4, cumulative threshold 12
- Special eligibility: US entities eligible to receive EU funding (reciprocity arrangement with NIH)
- Each proposal must target exactly one of the six designated virus groups listed in scope.
- Antibodies and antibody-derived proteins are explicitly excluded.
- Portfolio procedure: grants are awarded not only in ranking order but also to ensure coverage across the six virus groups
- Intellectual property clause: the granting authority may object to ownership transfers or exclusive licensing up to 4 years after project end
- No specific JRC involvement or clustering mentioned for this topic
Scientific range: what does the Commission expect from the HORIZON-HLTH-2027-01-DISEASE-05 grant?
What outcomes are expected?
At the conclusion of the project, the Commission hopes that a viable candidate antiviral agent will be available to researchers and clinicians to conduct more clinical studies or as an emergency response in case of an outbreak. It also desires to build upon the European portfolio of small-molecule antiviral drug targets in viral disease that is currently underrepresented. It does not request a couple of publications at the moment.
What is within scope?
Note: antibodies and antibody-based proteins are not covered under this. It can only be applied on small molecules.
- Identification and choice of a broad-spectrum antiviral candidate or of a candidate against variants.
- Optimization of SAR, including potency, selectivity, and pharmacokinetic properties of a candidate.
- In vitro antiviral activity and mechanism of action.
- In vivo animal validation, and in organotypic/organ models (in vitro).
- Preclinical testing in non-human primates should be demanded by regulatory authorities.
- Production of batches of the most promising candidates on GMP level.
- First-in-human clinical trials, with a regulatory road map.
- An intended scope can target either one of the six families of viruses. It is suggested that every proposal is dedicated to one family.
What are the specifically proposed research directions?
The research directions are strict and the work program permits minimal interpretation:
- Candidate drugs against one of the six groups of viruses: Junin/Lassa mammarenaviruses, tick-borne/Japanese encephalitis, Andes/Hantaan/Sin Nombre hantaviruses, Hendra, Enterovirus D68 or Venezuelan equine encephalite.
- Show intra-family activity (drug targets strains of virus closely related to the selected virus).
- Present a full preclinical data package (cell models, animal models and first-in-human safety trials).
- Consider regulatory issues in the proposal schedule and drug development plan.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-HLTH-2027-01-DISEASE-05?
Which scientific decisions are the most important?
- Wise selection of targets: Because a funded project is assured in each of the six groups (assuming all the proposals will pass the minimum quality criteria), selecting a less-competitive virus group may greatly increase the likelihood of the proposal receiving funding even with identical scientific quality. The same is usually true of portfolio-balancing funding calls.
- Evidence of a potentially viable regulatory pathway to first-in-human trials: Regulators need to be convinced of the existence of a viable regulatory pathway, and it is typically not enough to show that there is a clean mechanism of action and a robust in vitro data. The proposal should have a way of explaining the process to GMP manufacturing and regulatory engagement.
- Focus on SAR: The proposal call suggests the priority of the optimization of structure-activity relationships. When candidates are selected through phenotypic screening, mechanistic information must be supplied as an appendix; when the selection is made on a mechanistic basis, then the screening procedure needs to be explained.
- The breadth of action is an appropriate resource: Proposals that show activity against different strains of viruses, or even more generally, activity against different viruses (secondarily), is consistent with the preparedness objectives of the Commission. It is especially crucial in relation to the notion of intra-family and/or variant-transcending potential, which is outlined in the call.
- Demographic considerations: According to the work program, human clinical safety trials should take into account the differences according to sex, age, ethnicity, and disability.
- International cooperation: International cooperation is optional, but working with third-country institutions in endemic areas of the intended viruses provides substantial scientific validity and relevance to the study that extends beyond the geographic area covered.
Consortium & proposal-writing plan: what works best with this type of call?
- This type of RIA would typically involve a consortium of 8-12 partners; this may be expanded slightly if the clinical trials require very specialized knowledge.
- The consortium must contain researchers skilled in virology and medicinal chemistry/pharmacology, which cannot be found at one laboratory.
- The consortium should include a regulatory affairs specialist or an individual with experience in dealing with the EMA/FDA because regulatory interactions should be considered a part of the development phase and not an end.
- Examples: A typical and successful approach involves having an academic virology research group and a pharmaceutical/biotechnology partner with GMO manufacturing capability of the candidate drug.
- The call under the health cluster framework can be of great benefit to innovative SMEs capable of screening small-molecule antivirals and developing/optimizing candidates. The participation of the SMEs is clearly promoted, and the commercial potential of the successful candidate drugs is high.
- Third-country institutions within areas where the target viruses are endemic should be involved as scientific credibility partners, not to show endemic coverage.
- The proposal should be limited to a single target virus; any proposals presenting a number of virus groups will not get a positive review by the reviewers.
How would microfluidics contribute to this topic?
Traditional cell culture systems are insufficient when you want to know how a possible antiviral really acts in infected human tissue. 2D cell assays at rest provide you with a preliminary signal, but do not provide information on the interaction of the compound with the tissue environment, and permeation across barriers, and cell-cell interactions during infection. That is altered by microfluidic organ-on-chip setups.
- You wish to test the hypothesis, whether a candidate against the Hantaan virus truly protects the lung endothelium in the absence of off-target damage to the vascular wall. A lung-on-chip or vessel-on-chip model enables you to perform that experiment under conditions that are similar to human physiology, which cannot be completely replicated in a mouse model.
- The dynamics of infection at the tissue level. Microfluidic cultures can model the viral diffusion through epithelial or endothelial membranes, which is important for arenaviruses and hantaviruses that specifically infect epithelial or endothelial cells, respectively. Your consortium obtains information on the behavior of compounds in a more translatable context.
- Interstitial pharmacokinetics. In the case of small molecule antivirals, solving the problem of delivery of a compound to the right place at the right concentration is as crucial as activity in a test tube. Gut-on-chip or liver-on-chip models can assist you in characterizing early absorption, distribution, and metabolism and help you avoid using up the GMP budget on a candidate that will not behave in vivo.
- Sex and age effects of drugs. The call specifically requests that the biological factors such as sex be considered in the design of clinical safety. Organ-on-chip systems can incorporate hormonal environments and aged cell lines in ways inaccessible to animal models.
- Minimization of animal model reliance prior to the in-vivo studies required as a part of the work programme at least in the early rounds of screening. This is not only ethical but also accelerates the process of choosing candidates and saves on budget.
In your proposal, microfluidics can be classified as either in-vitro characterization or early in-vivo validation. The Microfluidics Innovation Center has been engaged in vascular, respiratory and barrier tissue systems that are directly pertinent to the infection biology of some of the six target virus groups. When your consortium is developing the preclinical package for a small-molecule antiviral candidate, this is where MIC can provide real value.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ – HORIZON-HLTH-2027-01-DISEASE-05
What is HORIZON-HLTH-2027-01-DISEASE-05 really about?
This call supports Research and Innovation Actions (RIA) to create innovative small molecule antiviral therapeutics against six priority groups of viruses that have epidemic potential. Each proposal must focus on a single virus group and aim to generate candidates eligible to enter first-in-human clinical safety trials by the conclusion of the grant.
Who can apply and how much funding is available per project?
The total budget is EUR 44.20 million for an average of 5 projects. Between EUR 9.00 and 11.00 million will be allocated to projects. US entities can also apply under a reciprocity arrangement with the NIH.
Which virus groups are in scope for this call?
These six identified groups of viruses include Junin/Lassa mammarenaviruses, tick-borne / Japanese encephalitis, Andes/Hantaan/Sin Nombre hantaviruses, Hendra virus, Enterovirus D68 and the Venezuelan equine encephalitis virus. The justification of one of the propositions targets one group only.
What is explicitly excluded from this call?
They do not include antibodies and antibody-derived proteins. This invitation is certain to small molecules. The intra-family or variant-transcending capability of the chosen virus group should also be demonstrated in each proposal.
How does the portfolio evaluation procedure work?
The levels of assessment include the following: Excellence 4, Impact 4, Implementation 4, and a total of 12. Besides the ranking, the Commission employs a portfolio process to ensure that at least one project is funded for each virus group, provided that the proposals meet all the thresholds.
What preclinical-to-clinical data does the Commission expect?
It will be mandatory to map the entire preclinical-to-clinical pathway: SAR optimization, in vivo characterization, in vivo animal studies, non-human primate testing, in case of regulatory requirements, GMO batch production, and first-in-human clinical safety studies, with a clear regulatory approval roadmap to market authorization.
What does an ideal consortium look like for this topic?
It is common to have a consortium of 8-12 partners, including academics with expertise in virology and medicinal chemistry/pharmacology, a manufacturing partner, and regulatory affairs experts. Involvement of innovative SMEs with small-molecule screening or optimization capacity is explicitly promoted and is commercially significant.
How does microfluidics fit into the preclinical work package?
One of the most significant advantages of organ-on-chip and microfluidic tissue models in the preclinical characterization phase is that they can be used to test antiviral compounds against virus-relevant tissue barriers (lung, vascular, and endothelial) under physiological flow conditions to provide translatable data that cannot be obtained with a static 2D assay.
What third-country and demographic requirements apply?
Epidemiological relevance is provided with the encouragement of third-country partners in the regions of endemicity of target viruses. The regulatory bodies should also be engaged at the earliest stage of the project. The work program requires the clinical safety study design to account for sex, age, ethnicity, and disability.
What are the IP conditions specific to this topic?
The granting authority also has the power to object to any relocation or exclusive licensing of the results within a span of 4 years after the end of the action, as per the specific Section in the Grant Agreement of Annex 5. The consortium partners and the SMEs should take this into account from the very start when planning how to exploit it in their strategies. Check the Funding and Tenders Portal for more information.
