Tips & Tricks for a successful HORIZON-CL3-2026-01-FCT-06 proposal
Opening
06 May 2026
Deadline
Keywords
synthetic biology
RIA
bioterrorism
gene-editing technologies
DIY biohacking laboratories
civil security
EGNOS
data management
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
HORIZON-CL3-2026-01-FCT-06: Prevention and mitigation of misuse of synthetic biology for bioterrorism purposes
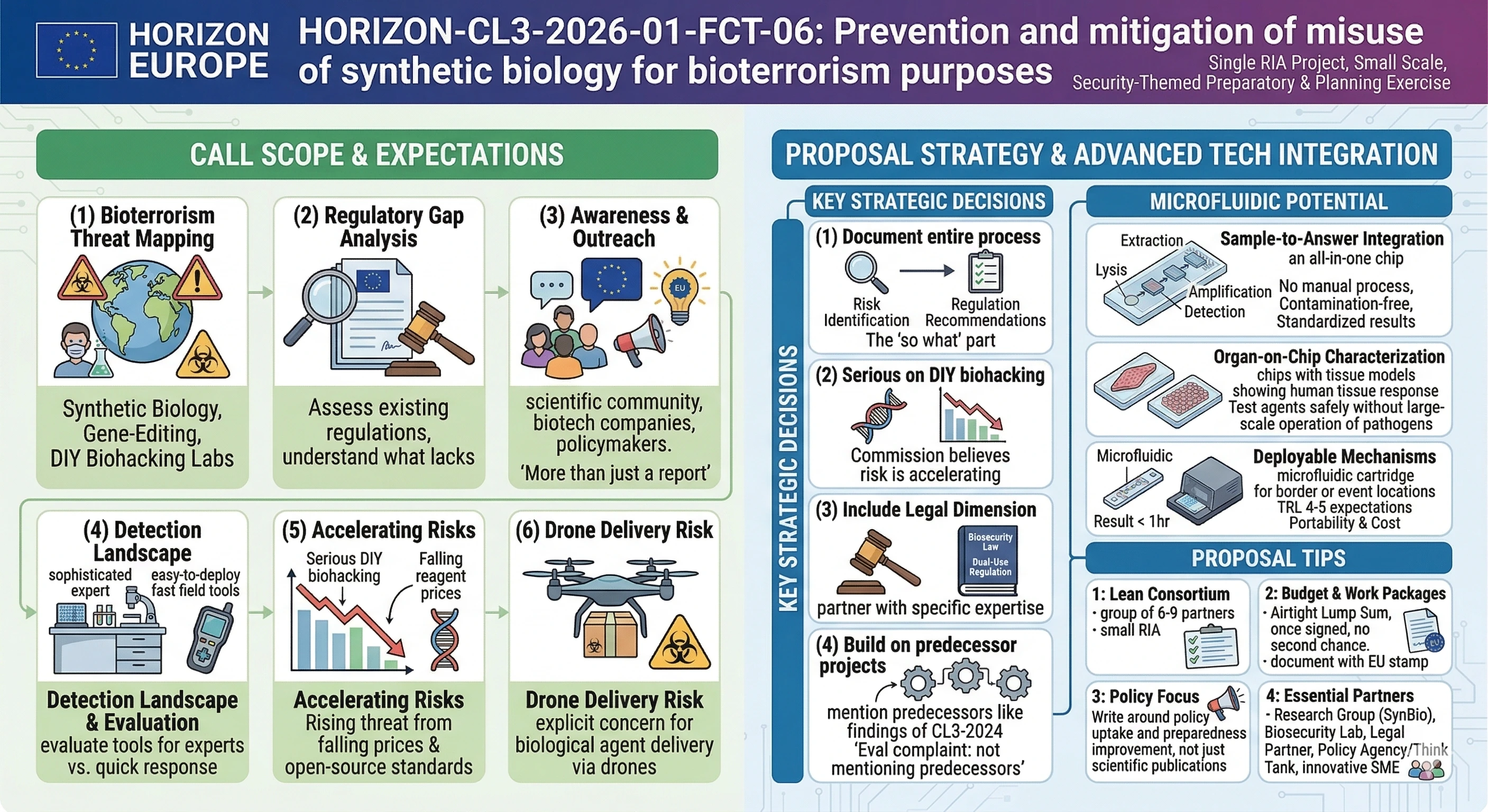
The Commission desires a consortium of one to map the bioterrorism threats posed by increasing access to synthetic biology, gene-editing technologies, and DIY biohacking laboratories. This does not imply the call to construct a detection platform. What they seek is a risk assessment, a regulatory analysis and awareness resources to the research fraternity. Consider it as a preparatory and planning exercise that is security-themed and which is financed as a single RIA at a reasonably small scale.
Discover more!
Administrative facts: what do we know about the HORIZON-CL3-2026-01-FCT-06 call?
Which call is it, and when is the opening and the deadline?
● Call name: Civil Security for Society 2026
● Call identifier: HORIZON-CL3-2026-01
● Destination: Better protect the EU and its citizens against Crime and Terrorism
● Topic: HORIZON-CL3-2026-01-FCT-06
● Opening date: 06 May 2026
● Deadline: 05 November 2026 (17:00 Brussels time)
● Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
● Overall topic budget: EUR 3.00 million
● Number of projects expected to be funded: 1
What are the key eligibility and evaluation conditions?
● Standard RIA evaluation thresholds apply (General Annex D).
● Lump sum grant. Costs take the form of a lump sum, not actual costs.
● Target TRL: activities are expected to reach TRL 4 to 5 by the end of the project.
● Legal entities established in China are not eligible for RIAs under this destination.
● Security sensitive: some activities may involve classified background or produce EUCI/SEN results. Worth checking twice before you plan your dissemination.
● If the project uses satellite data, Copernicus and/or Galileo/EGNOS must be used.
● No specific minimum number of Police Authorities required (unlike several other FCT topics), but engagement with the Europol Innovation Lab is expected.
● Gender dimension should be addressed only if relevant.
Deadlines of European Programmes 2026/2027
Get the MIC Horizon Europe 2026/2027 Calls Calendar:
-All Horizon Europe deadlines (by cluster and call).

Scientific range: what does the Commission expect from the HORIZON-CL3-2026-01-FCT-06 grant?
It is broader than pure bio-detection. The Commission believes that this is a high-impact, low-probability threat, and is interested in a project that provides insight and readiness as opposed to a complete product.
● Risk mapping: examine the existing and emerging bioterrorism threat of synthetic biology, gene editing and increased accessibility to commercial DNA synthesis services and open databases.
● Regulatory gap analysis: understand what lacks in European security regulations on the subject of the masses in this regard. What activities should be monitored with more force, what work at present goes through the cracks.
● Detection technology landscape: evaluation of both sophisticated detection tools in expert laboratories, and the easy-to-deploy techniques of fast field intervention. The length of incubation of biological agents is cited in the call as a specific issue.
● Drone weaponisation: the work programme explicitly raises that the increased application of drones might be extended into the field of biological agent delivery. This should not be neglected in your proposal.
● Awareness and outreach: scientific community, the biotech companies, and the policymakers must be aware of how synthetic biology research can be abused. At least we have found that reviewers would prefer a report stored on a shelf to have tangible plans on how we can carry out awareness campaigns.
Regarding anticipated results, the Commission desires a better evidence base among policy makers and practitioners in the security field and increased awareness among scientists in synthetic biology. Output in publicity is secondary. Operational utility and policy preparedness is what counts.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-CL3-2026-01-FCT-06?
Which scientific decisions are the most important?
● Document the entire process of risk identification up to regulation recommendations. Then, do not end at the threat assessment. The so what part will be sought by reviewers.
● Be serious with the DIY biohacking scene. On-campus laboratories, falling prices of gene sequencing reagents, open source standards. Here the Commission believes the risk is accelerating.
● Include a legal dimension. The call makes it very clear regarding a legal analysis of monitoring needs. Having a partner with experience in the area of biosecurity law or dual-use regulation would add a lot of power to your consortium.
● Build on the findings of HORIZON-CL3-2024-FCT-01-04. The work programme states so forthright. The most common evaluator complaint is not mentioning predecessor projects (we have tripped up more than one consortium with this particular complaint).
● Do not overengineer the detection part. TRL 4–5 informs you that the Commission is not anticipating a field-deployable product. Proof of concept is enough.
● Respond to drone-based biological delivery cases. Even briefly.
● Think through engagement with the Europol Innovation Lab not at the end of the month 30.
Consortium & proposal-writing plan: what works best with this type of Security RIA?
● Since there is only one project with EUR 3 million, maintain the lean consortium. Probably not more than six or nine partners. The lump sum distribution is cumbersome with too many partners in a small RIA.
● You must have at least: a research group in synthetic biology, a biosecurity or defence laboratory, a legal and regulatory partner and a policy think tank or public health agency. Better still, you can include a security practitioner or national CBRN authority.
● Technical credibility and exploitation potential would be introduced with an innovative SME with expertise in biosensing or bio-detection. In a low-TRL project, evaluators can see the pathway between research and market.
● Considering the fact that this issue can yield security-sensitive outcomes, consider restraining your consortium agreement and data management at the very first step taking into consideration the EUCI/SEN restriction.
● Lump sum format translates to the fact that the work packages and your budget distribution must be airtight when submitted. Once the grant is signed there is no second chance.
● Write around policy uptake and preparedness improvement, not scientific publications. The Commission is purchasing preparedness not documents.
How would microfluidics contribute to this topic?
Traditional methods of detection of biological agents are often based on centralised laboratory systems, slow culture-based systems or costly mass spectrometry systems. All of them are not fast enough with a limited time frame to respond. Microfluidic systems squeeze whole assay processes on a miniature chip, and that alters the formula of quick bio-threat detection.
● Suppose that you have to test an environmental sample of engineered pathogens or synthetic toxins at a border or an event location. A microfluidic cartridge with a multiplexed nucleic acid amplification assay can provide a result in less than an hour, even in the absence of trained lab personnel. That is the type of deployable mechanism the call text is seeking.
● Microfluidics excels in sample-to-answer integration. Lysis, extraction, amplification, detection, everything in one. It requires no manual pipetting, no contamination possibility, and will always give the same results.
● In the case of the expert lab side, organ-on-chip models may be utilized to investigate the behavior of introduced biological agents with human tissue, and could be used to help characterize the threats without operating with live pathogens at scale.
● Portability and cost. The prototype of a microfluidic biosensor is in the range of TRL 4–5 expectations and can be produced at volumes that are equivalent to preparedness stockpiling scenario.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
