Tips & Tricks for a successful HORIZON-HLTH-2026-01-TOOL-03 proposal
Opening
10 February 2026
Deadline
Keywords
Cluster health
New Approach Methodologies
RIA
regulatory testing
iPSC models
biomedical research
population variability
AI predictive models
NAMs
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
HORIZON-HLTH-2026-01-TOOL-03: Integrating New Approach Methodologies (NAMs) to advance biomedical research and regulatory testing
The European Commission would be concerned with initiatives that would achieve full penetration of the New Approach Methodologies across the entire biomedical research and regulatory testing pipeline. At the early discovery phase, since its inception, translation of medicinal products, clinical approval of drugs, regulatory approval of medical devices, and regulatory approval of chemicals.
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2026-01-TOOL-03 call?
Which call is it, and when are the opening and the deadline?
- Call name: Cluster 1 – Health (Single stage – 2026)
- Call identifier: HORIZON-HLTH-2026-01
- Destination: Developing and using new tools, technologies and digital solutions for a healthy society
- Topic: HORIZON-HLTH-2026-01-TOOL-03 – Integrating New Approach Methodologies (NAMs) to advance biomedical research and regulatory testing
- Opening date: 10 February 2026
- Deadline: 16 April 2026
- Type of Action: Research and Innovation Actions (RIA), 100% funding rate
What about the budget and estimated size of the project?
- Overall topic budget: EUR 49.00 million
- Indicative number of funded projects: 7
- Budget per project: EUR 5.00 to 8.00 million
What are the key eligibility and evaluation conditions?
- Award thresholds: Excellence 4/5, Impact 4/5, Implementation 4/5, cumulative 12/15
- Chinese entities are not eligible for this RIA
- US entities are eligible for EU funding
- JRC may join after selection as beneficiary (zero funding) or associated partner
- All funded projects must form a joint cluster and dedicate \~2% of budget to networking activities
Deadlines of European Programmes 2026/2027
Get the MIC Horizon Europe 2026/2027 Calls Calendar:
-All Horizon Europe deadlines (by cluster and call).

Scientific range: what the Commission expects from the HORIZON-HLTH-2026-01-TOOL-03 grant?
Despite more than two decades of being in the spotlight, NAMs have very little integration in the regulatory field due to the assistance of the European Union. This call is intended to alter this. The areas of technology are:
- In-vitro and human ex-vivo experimental methods.
- Organ-on-chip and Organoids (OoC) systems.
- Human tissue models
- Induced Pluripotent Stem Cell (iPSC) has been used.
- Virtual twin tools and in-silico computational tools.
- Artificial intelligence predictive modelling.
The proposals must create or capitalize on scalable, repeatable platforms on the basis of:
- Advanced in-vitro assays
- iPSC-based models Patient or healthy donor-based Organoid or complex OOCS.
- Cloning of human physiological and pathological tissues was done.
Application tracks are of two kinds:
- In biomedical research platform: the platforms must improve the quality of the disease modelling in case there is no human relevance of the animal models.
- In the regulation of chemicals, medicinal products and medical devices, the proposal must provide a clear context of use. The validation plans would be in accordance with OECD and/or EMA. Close communication with regulators should be maintained at an early stage.
Some other expectations are also mentioned in the work programme:
- In particular, the incorporation of embedded sensors for real-time physiological monitoring is encouraged.
- Biological diversity should be considered, as well as variations in genetics, phenotype, age, immune condition and microbiome.
- The promotion of complementarity of approaches, including the deployment of AI-based predictive modeling or the creation of virtual twin technology.
- The data will have to comply with the FAIR principles and be linked to European Health Data Space (EHDS) or ESFRI research infrastructures.
- The participation of the JRC and EURL ECVAM is desirable, namely, the standardization/validation of in-vitro methods.
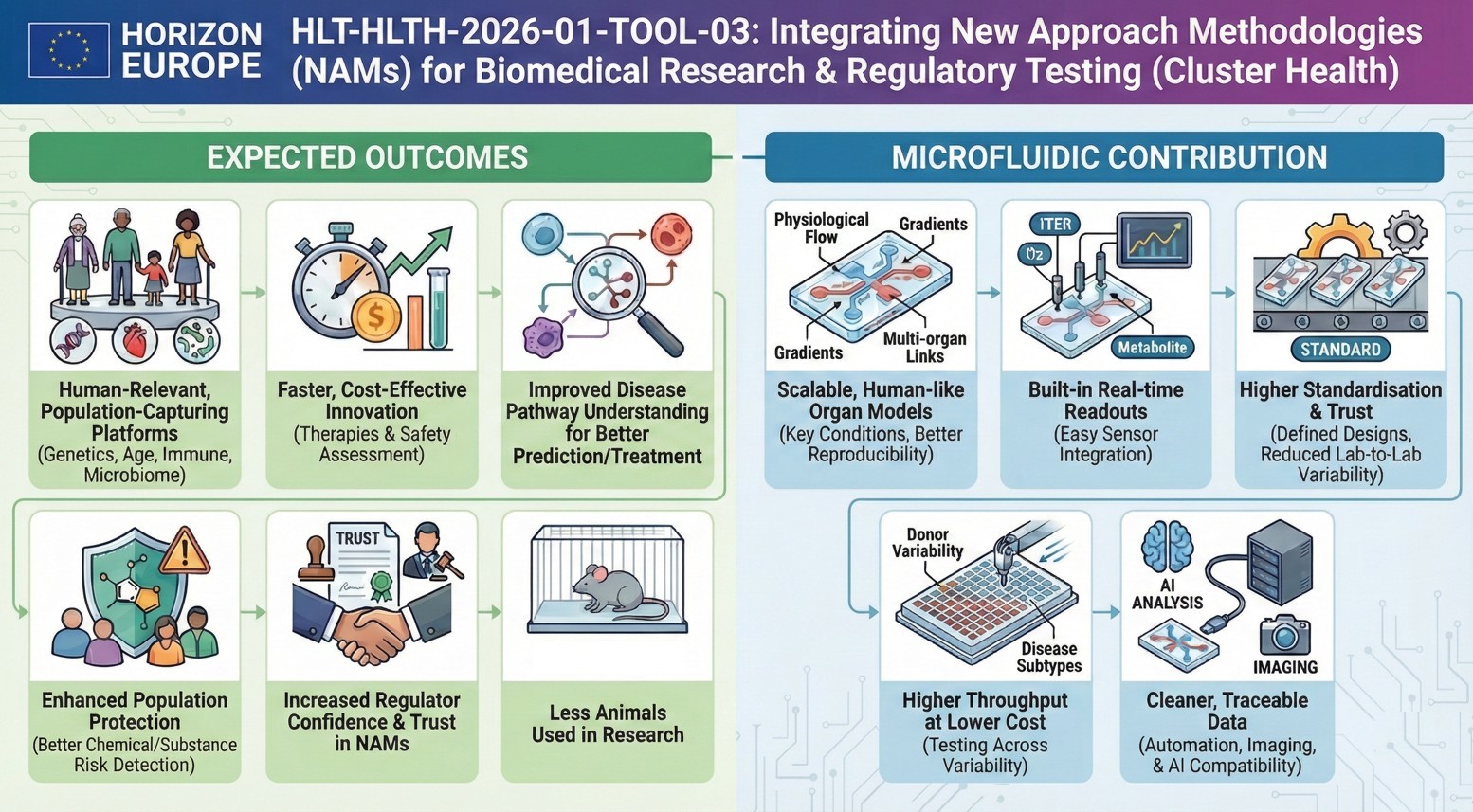
The call is phrased in the format of the five expected outcomes:
- Less expensive, faster platforms are made available to industries.
- Patients benefit by having enhanced disease prediction and treatment.
- The citizens are safeguarded against environment and chemical dangers.
- NAMs increase the trust of the regulators.
- Less live animals are utilized in research and testing
Scientific strategy: How can you enhance your chances of being funded through HORIZON-HLTH-2026-01-TOOL-03?
Which scientific decisions are the most important?
- Target organ-on-chips and organoids. These are especially referred to as core technologies. These proposals are supported by the fact that there are various organs that are connected with each other and patient-specific models.
- Embed real-time sensing. Embedded sensors are specifically requested in the work programme. The assessors will take note of how this reacts on you or not.
- Capitalise on the problem of population diversity. Use cells of various donors with various genetic background, age, sexes, and immunological contents.
- Combine wet-lab with AI. One possible example of meeting the demands of a multitude of calls is multi-purpose pipelines in which machine learning models (that run on the data that is supplied by OoC models) give feedback on the data.
- Consult the regulators at a very early stage. Included EMA letters of support, national letters of support or OECD letters of support. Evaluators will not be moved by promises which are generic.
- Normalize validation, make validation conform. See OECD test guidelines or EMA qualification pathways.
- Design a tangible EURL ECVAM partnership. This will demonstrate that you are not imagining plans of translating something, after all.
Consortium & proposal-writing plan: what works best with this type of Health RIA?
- RIA of 5 to 8 million EUR will comprise of 8-15 partners with 5-6 EU or Associated Countries.
- The stakeholders within the academic field, the health facilities, SMEs, industries and regulators specifically have to be involved in the work programme. Evaluators will verify this.
- It should be scientifically supported by leading academic laboratories working on NAMs (OoC, organoids, iPSC, in silico).
- Translational trustworthiness is associated with the existence of a partner in the pharma / medtech industry.
- The validation pathway is empowered by the presence of an expert in regulatory science with experience in EMA or OECD.
- Clinical partners become essential when the work involves patient-derived cells or clinical validation steps.
- An innovative SME is to be taken into consideration. The already established companies involved in the business of manufacturing Organ-on-Chip products and microfluidic biosensors or drug discovery artificial intelligence, or CRO outsourcing services are coming in with business awareness and realistic strategy of exploitation. The Commission values them in transforming the research into marketable products.
- Identify the language of the anticipated outcomes and project it onto the work programme on your proposal.
- Be explicit about your context of use in the regulations.
- Provide a pathway between the platform and end users through letters of intent.
- Bring the 3Rs narrative (replace, reduce, refine animal testing) to actuality, which is one of the greatest policy driving forces in this appeal.
- Address sex and gender considerations thoughtfully.
- Turn the clustering work package into reality and not an act.
How would microfluidics contribute to this topic?
Among the most popular technologies to be listed in the work programme, so-called Organ-on-Chip systems should be mentioned that, in fact, are microfluidic devices. They use microchannels, which are controlled to replicate physiological flow, nutrient delivery, mechanical stimuli and inter-organ communications. In this case only microfluidics cannot be used. It lies in the middle of what the Commission is demanding.
In addition to this basic functionality, microfluidics realizes some of the privileges that are directly associated with the needs of the call:
- Application of microfluidic platforms makes OoC diversity possible. In contrast to the traditional well-plate-based platforms, the cells of various donors are tested simultaneously in the microfluidic platforms, which are significantly smaller in size.
- The integration of AI uses the ongoing and high-resolution streams of data offered by sensor-integrated microfluidic platforms and, therefore, allows this novel technique as suggested by the commission through its work program.
- Microfluidic platform parallelization can also be used to reduce the cost of reagents and can scale up to simultaneously test more conditions in high-throughput screening assays.
For any consortium planning to apply for the HORIZON-HLTH-2026-01-TOOL-03 call, working with a microfluidics expert, either in a university lab or in an innovative SME in OoC or biosensor technology, is a very simple way to align with the scientific priorities set for this work program.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ - HORIZON-HLTH-2026-01-TOOL-03
What is HORIZON-HLTH-2026-01-TOOL-03?
It is an RIA call that focuses on the implementation of New Approach Methodologies (NAMs) in biomedical research and regulatory testing. The objective is to replace animal testing with human-relevant platforms throughout the entire innovation pipeline, including early discovery and regulatory evaluation of medicines, medical devices, and chemicals.
Deadline and budget of the call?
The call will be opened on 10 February 2026 and will have a deadline of 16 April 2026 17:00 Brussels time. The overall budget is EUR 49 million, which is likely to finance about 7 projects, each with a budget of EUR 5-8 million. Refer to the fact that the opening can be changed by +-1 month and the deadline can be extended by 2 months.
What are New Approach Methodologies (NAMs)?
The NAMs include human-relevant methods such as complex in vitro and ex vivo models, iPSC-based models, organoids, organ-on-chip models, human tissue models, AI predictive modeling, virtual twin technology, and embedded real-time monitoring and sensing. It is interested in methods that are more realistic and relevant to human physiology and pathology than traditional animal-based models.
What are the most important anticipated outcomes?
Projects are expected to provide human-relevant systems that include population variability, accelerated and cost-efficient innovation, enhanced disease pathway knowledge, improved chemical risk detection, enhanced regulatory confidence on NAMS, and improved animal use in research and test. More importantly, it must be shown that the results are not only scientifically excellent but also accepted by regulators.
What is the requirement of cluster approach?
Each and every funded project must be part of a compulsory cluster that involves joint activities, common dissemination, synchronized reporting, and collaborative standard-setting. The suggestion should also plan and budget for cluster participation, as this is not an optional feature.
Why is a consortium competitive in this topic?
Powerful consortia include academia (mechanistic depth), technology developers (platforms and sensors), research infrastructures (harmonization and standards), and regulators or reference laboratories (acceptance pathways). The involvement of industry is essential, but must extend beyond end-user involvement to define performance requirements and the impediments to adoption. The credibility of exploitation is enhanced by incorporating innovative SMEs with technologies that enable NAM.
What should be proposed in regard to regulatory acceptance?
Do not consider the regulation interface as a mere dissemination. Identify early context of use (what decision, to whom, under what conditions), organize interactions with regulators and reference bodies, and demonstrate how regulatory feedback will influence methods and evidence packages. Incorporate articulate multi-site replication plans that indicate repeatability, reproducibility, and transferability.
What are the essential data and compliance issues?
The proposals need to operationalize FAIR principles of data using tangible standards, metadata, repositories, and access models. demonstrate compliance with GDPR requirements, adherence to EU health data approaches (EHDS is mentioned), and compatibility with appropriate ESFRI infrastructures. Present data governance, which is perceived to be EU-native, and not generic compliance statements.
What is the role of population variability in proposals?
The variability of the population should not be an afterthought; it should be built in. The proposals must cover the diversity of donors, age and sex factors, immune systems, microbiome, and exposure conditions. Such variability can be investigated cost-effectively using high-throughput methods, such as parallelized microfluidic arrays.
What is the application of microfluidics in this topic?
Microfluidics enables controllably perfused, gradient-based, and multi-organ-coupled organ-on-chip systems to be more human-relevant. They inherently incorporate sensors to monitor in real time, minimize inter-labor differences through uniform protocols, support large-scale population investigations, and generate clean data for AI analysis. These features specifically address the issues of reproducibility, scalability, regulatory credibility, and real-time physiological measurement, as emphasized in the topic.
