Tips & Tricks for a Successful HORIZON-CL6-2026-01-CIRCBIO-07 Proposal
Opening
17 April 2026
Deadline
Keywords
Bio-Based Innovation
Biotechnology Solutions
Biomanufacturing Processes
Sustainable Bioeconomy
Industrial Biotechnology
Circular Bio-Based Systems
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
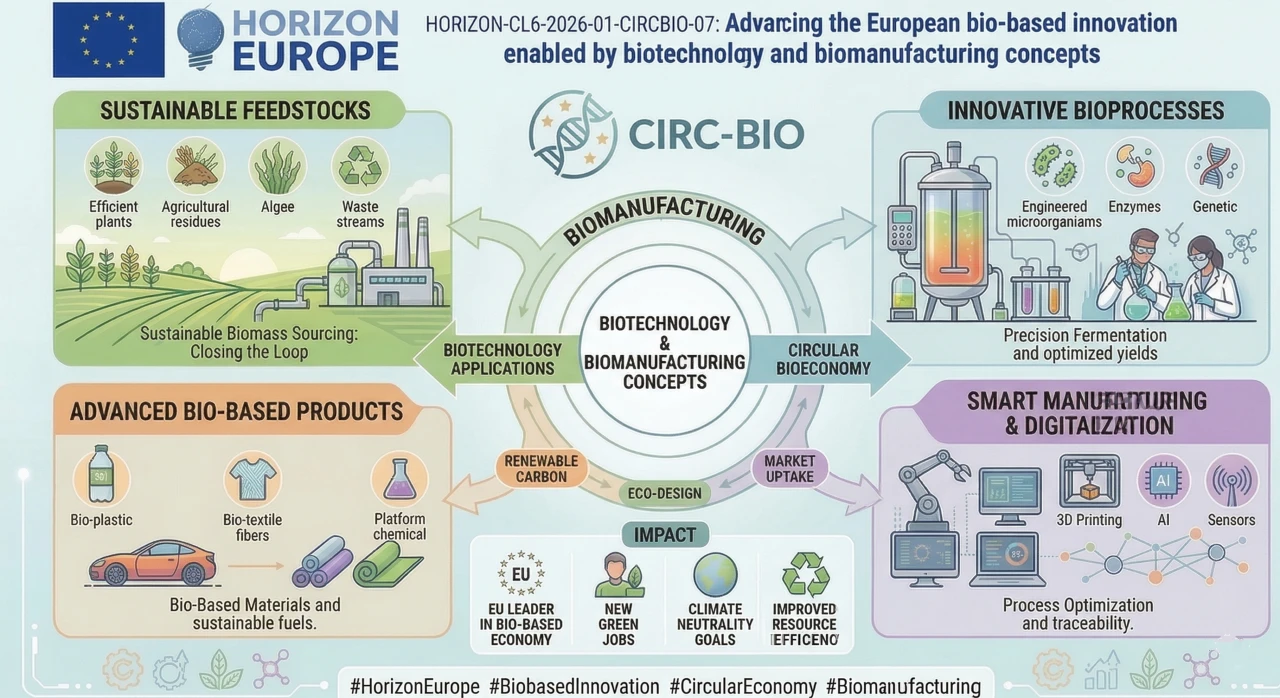
HORIZON-CL6-2026-01-CIRCBIO-07: Advancing the European bio-based innovation enabled by biotechnology and biomanufacturing concepts
The Commission would like to see biotechnology and biomanufacturing leave the bench of the lab and come into the industry that can be used. Not tomorrow, not at scale but at least to a credible demonstration of reality. The concept is to create bio-based processes, products or materials that can be utilized in the circular value chains and reduce environmental footprint. Consider synthetic biology, gene engineering, metabolic engineering, microbiome tools. All it was related to digital and was intended to substitute fossil-based methods.
Discover more!
Administrative facts: what do we know about the HORIZON-CL6-2026-01-CIRCBIO-07 call?
Which call is it, and when is the opening and the deadline?
● Call name: Horizon Europe Work Programme 2026-2027, Cluster 6, Food, Bioeconomy, Natural Resources, Agriculture and Environment
● Call identifier: HORIZON-CL6-2026-01
● Destination: Circular economy and bioeconomy sectors
● Topic: HORIZON-CL6-2026-01-CIRCBIO-07
● Opening date: 17 April 2026
● Deadline: 17 September 2026
● Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
● Overall budget for this topic: EUR 12.00 million
● Number of projects expected to be funded: 3
● Estimated EU contribution per project: around EUR 4.00 million
● Lump sum funding applies
● TRL target: 4 to 5 by the end of the project, starting from any TRL
What are the key eligibility and evaluation conditions?
● Standard Horizon Europe eligibility and evaluation criteria apply (General Annexes A through G)
● No JRC participation mentioned for this topic
● SSH disciplines and SSH experts are required, not optional
● DNSH (Do No Significant Harm) principle must be respected
● Data must be FAIR (Findable, Accessible, Interoperable, Reusable)
● Cooperation between all funded projects under this topic is mandatory, with dedicated tasks and budget
● Synergies encouraged with CIRCBIO-10 (bio-based innovation in society) and microbiome or CCU topics from this work programme
Deadlines of European Programmes 2026/2027
Get the MIC Horizon Europe 2026/2027 Calls Calendar:
-All Horizon Europe deadlines (by cluster and call).

Scientific range: what the Commission expects from the HORIZON-CL6-2026-01-CIRCBIO-07 grant?
Fundamentally, this is the subject of driving key enabling biotechnologies up the readiness ladder. Synthetic and molecular biology, gene editing, metabolic engineering, applications of the microbiome, and biofoundry approaches are the playground listed by the Commission. What specifically is not allowed: health biotechnology and biofuels. This is not your thing so should your idea reside in pharma or energy.
At the beginning, it is supposed to be digitally integrated. It implies AI, machine learning, bioinformatics embedded into the R&D process. Not as a side deliverable. As a design choice.
In addition to the technology, the work programme requires convergence in biotechnology and nature based solutions. Carbon capture, wildlife conservation, environmental cleaning. The Commission would like to have an assurance that your bio-based innovation does not only work in a laboratory environment but also relates itself to actual ecological concerns.
Policy recommendations should also be provided by the projects. And mobilise the civil society, NGOs, consumer organisations. The phrasing is still open to interpretation, although what evaluators will be seeking is actual engagement with the stakeholders, and not a token dissemination event in month 36.
The anticipated results narrow down to two elements, that is, improve the EU competitiveness in bio-based innovation, and show quantifiable environmental benefits in terms of climate, biodiversity, and resource efficiency.
Scientific strategy: How can you enhance your chances of being funded through HORIZON-CL6-2026-01-CIRCBIO-07?
Which scientific decisions are the most important?
● Select one or two enabling technologies and dive. Trying to cover synthetic biology, gene editing, microbiome, and biofoundries simultaneously in a EUR 4 million project will not persuade the evaluators that you are capable of performing at TRL 4-5.
● Validation on the pilot scale is promoted. Your proposal could be losing to a rival who intends to have pilot run, even in a small scale, if your proposal remains absolutely at the bench level.
● Demonstrate the integration online. An AI bolted at the end of the work seems to be an afterthought, a work package. Integrate bioinformatics or ML in the experimental design since the first month.
● Convergence on nature solutions. This is not decoration. We have already encountered reviewers coding proposals where NbS is presented as an individual add-on to a research question, but not as part of it.
● Handling safety and risk evaluation directly in gene editing or synthetic biology work. It is explicitly mentioned in the text of the call.
● Make your environmental sustainability pitch with figures. Semi-promises of a smaller footprint will not do (do check your LCA plan twice before handing it in).
● Policy recommendations are obligatory. Allocate real effort to this. The Commission is not after a two-page deliverable in month 48.
Consortium & proposal-writing plan: what works best with this type of Health RIA?
● Strive to have a number of partners of eight or twelve. EUR 4 million in lump sum is not so much when you have to divide it between fifteen people, and the administration cost of lump sum with excessive number of beneficiaries is a pain.
● You require high-quality biotech or life science laboratories, most likely two or three, and reputations in the particular technologies you are going after.
● The section impact and the exploitation plan are enhanced by an innovative SME with the actual biomanufacturing or bioprocess capability. The call specifically appeals to the input of SME as beneficiaries or external actors.
● Have at least one partner who has environmental or ecological background for NbS convergence dimension. Many biotech consortia fail in this regard.
● In case you can introduce a new member to Cluster 6, make it clear. It is mentioned in the text of the work programme and noticed by reviewers.
● SSH expertise is mandatory. Do not make this a box to be ticked. When a social scientist contributes towards the perceived and acceptance work package by the people, he or she adds real value.
● In writing: the lump sum format implies that your budget story must be tight and internally sound. Reviewers are aware of the actual price of a pilot campaign. When your figures do not add up, then the entire lump sum set up is a weakness.
● There is one task that should be devoted to collaboration with other projects funded in this topic. Plan it out correctly in the beginning.
How would microfluidics contribute to this topic?
The traditional screening and characterisation biotech processes are slow. They use large quantities of reagents and biological sample and when you want to test hundreds of enzyme variants or microbial strains, the bottleneck is the throughput of your experimental system.
● Microfluidic systems allow you to effectively screen thousands of variants of an enzyme or strain in parallel, through droplet-based assays that can be run on microlitre volumes. Imagine you are designing a metabolic pathway to a novel bio-based monomer: you can measure quantities of expression in hundreds of different conditions in an afternoon rather than the weeks on the bench.
● In gene editing processes, microfluidics allows delivery of the forms of CRISPR to specific cells and sorting phenotypes in real time. Same compound, different host, different outcome all monitored in real time.
● The biofoundry solutions are based on miniaturisation and automation. Microfluidic modules are designed to fit directly into biofoundry pipelines and process cell culture, selection and analytics within one chip.
● It is now possible to screen environmental fate on-chip: bio-based materials can be subjected to simulated environmental conditions at the small scale and kinetics of degradation measured. Early information on sustainability is provided to your consortium without the need to wait until large-scale field tests can be carried out (this one is significant to the NbS convergence the call requests).
● The safety assessment aspect can also be supported with the help of organ-on-chip and tissue models in case your project is based on new bio-based materials that require characterisation in terms of toxicology.
Microfluidics provides your proposal with an actual response to the throughput, resource efficiency and digital integration needs that run through this call. Partnering with a microfluidics firm, especially an innovative SME with application-ready platforms, is a sign to the evaluators that your consortium would be capable of delivering the validated results within the project timeframe and the lump sum budget.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
