Tips & Tricks for a successful HORIZON-HLTH-2027-02-IND-02-two-stage proposal
Opening
10 February 2027
Deadline
Keywords
Portable & versatile
WHO REASSURED
rapid testing
Point-of-care diagnostics
real-time connectivity
IVDR / MDR
Sample-to-answer
user-friendly
AI/ML
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
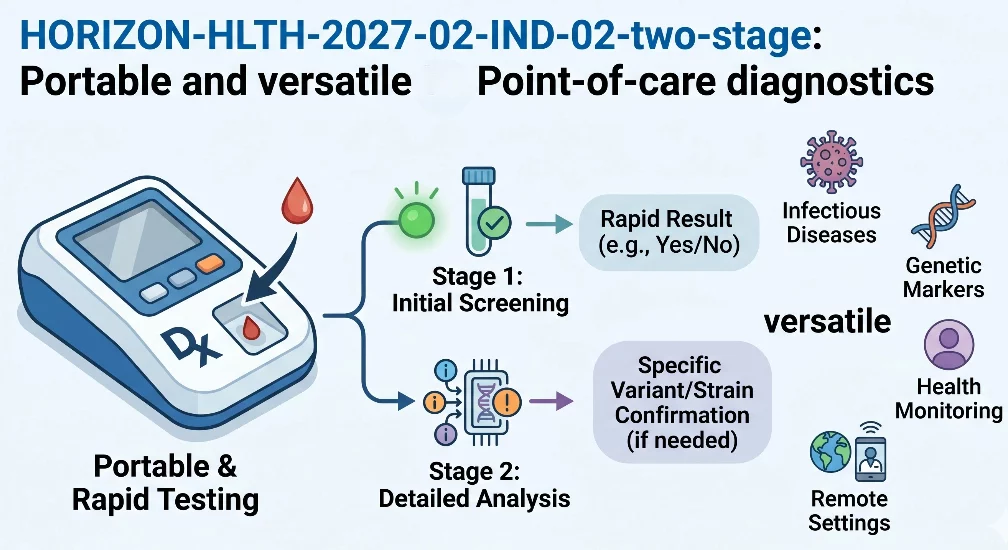
HORIZON-HLTH-2027-02-IND-02-two-stage: Portable and versatile point-of-care diagnostics
The point-of-care testing has evolved. Lab-based tests are nearly as accurate as their application in the field. However, it cannot be adopted on a large scale clinically by almost everyone, and the Commission is aware of that. This is a call to bridge the remaining gap: fake results, sloppy sample preparation, and machines that cannot withstand the rigors of a well-equipped hospital. It is not about increased research on PoC potential. It is devices that are practical, meet EU regulations, and demonstrate their worth in actual clinical practice.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2027-02-IND-02-two-stage call?
Which call is it, and when is the opening and the deadline?
- Call name: Cluster 1 – Health (Two Stage – 2027)
- Call identifier: HORIZON-HLTH-2027-02-two-stage
- Destination: Maintaining an innovative, sustainable, and competitive EU health industry
- Topic: HORIZON-HLTH-2027-02-IND-02-two-stage
- Opening date: 10 February 2027
- Deadline: First Stage: 13 April 2027 (blind evaluation)
- Second Stage: 22 September 2027
- Type of action: Innovation Action (IA)
What about the budget and estimated size of the project?
- Total indicative budget for the topic: EUR 39.30 million
- Expected number of funded projects: 6
- Expected EU contribution per project: EUR 5.00 to 7.00 million
- Costs will be paid in a lump sum.
What are the key eligibility and evaluation conditions?
- First-stage threshold: 4 (Excellence) and 4 (Impact); proposals admitted to stage 2 must collectively represent roughly four times the available budget
- Second-stage threshold: 4 (Excellence), 4 (Impact), 4 (Implementation); cumulative threshold of 12
- First-stage proposals submitted under blind evaluation: no organization names, acronyms, logos, or personnel names in abstract or Part B at stage 1
- US entities are eligible under this topic (in recognition of NIH reciprocity)
- Entities controlled directly or indirectly by China are NOT eligible.
- Satellite data, if used: Copernicus and/or Galileo/EGNOS mandatory
- Transfer of ownership or exclusive licensing of results: granting authority may object up to 4 years after project end
Scientific range: what does the Commission expect from the HORIZON-HLTH-2027-02-IND-02-two-stage grant?
What outcomes are expected?
The Commission desires deployable devices, but not prototypes in a laboratory. A PoC tool that actually accelerates the treatment decisions should be available to the healthcare professionals at the end of the project. The patients receive quicker and more precise diagnoses. In its turn, health systems receive improved data in disease control. That is what the Commission wants funded projects to show, end-to-end.
What is within scope?
- PoC diagnostic devices against infectious as well as non-communicable diseases.
- Optimization of the device in regard to the WHO REASSURED requirements: real-time connectivity, simple to collect specimens, affordable, sensitive, specific, user-friendly, fast, sturdy, minimal equipment requirements, and end-user deliverability.
- Sample-to-answer (Complete workflow, no lab in-between steps)
- ML/AI algorithms to analyze, recognize, and read the data.
- Mobile phone acquisition, readout, or sample-processing components.
- Biocompatible materials that can be mass-produced.
- The high-throughput configurations were applicable.
- Plan on resource-constrained grounds (poorer countries, regions with poor health infrastructure)
The call does not finance individual feasibility or low-level research. In the absence of clinical validation and a path to EU regulatory compliance (IVDR or MDR), the proposal falls out of scope.
What are the specifically proposed research directions?
These are not optional activities, but necessary ones, called required ones in the work program:
- Optimization of the targeted PoC device(s) with respect to a specified clinical needs assessment that encompasses all care pathways and system-level perspectives (not only clinician input).
- A direct comparative study that shows quantifiable performance improvements over the existing standard-of-care testing.
- IVDR or MDR Clinical studies in the feeding of conformity assessment.
- In the case of infectious disease, methods can differentiate between viral, bacterial, or fungal sources.
- When using non-communicable diseases, preference should be given to emergency cases where the results are life-saving.
- Not as an afterthought, but rather the integration of Health Technology Assessment into the device’s development.
Scientific strategy: how can you enhance your chances of being funded through HORIZON-HLTH-2027-02-IND-02-two-stage?
What scientific choices matter most?
- Have the basis of all that in a clinical needs assessment. The Commission is categorical. The evaluation should not be limited to a clinician survey; it should encompass the entire care pathway. This will be checked by the evaluators.
- Your checklist is the REASSURED framework. Discuss every requirement in the proposal and clarify which features your device supports and what you are working on. Leaving this out is an interpretation of not reading the call.
- Choose a particular area of disease and remain in it. We have had proposals to cover both infectious and non-communicable diseases on the same machine. This is likely to be perceived by evaluators as scope-spreading, rather than versatility.
- Design regulatory compliance Day one. Conformity to IVDR or MDR is not a deliverable added at the end. Early mention of notified body contacts, definitions of intended use, and risk classification. The projects that make regulation a post-project issue hardly pass stage 2 review.
- The value-based concept is not a choice. Use of HTA as a direct mention and indicate HTA bodies that are applicable to your target market.
- The incorporation of AI should be justified. In the case of including ML/AI components, what does it detect that human analysis fails to detect? Claims about AI being powered are insincere and cannot withstand expert scrutiny.
- Limited settings involving resources are not a luxury, but a necessity. The Commission declares this directly. If your device can operate in low-infrastructure settings, state that in the impact section rather than in an annex of the technical section.
Consortium & proposal-writing plan: what works best with this type of call?
- The Action of Innovation, not RIA. 8 to 12 partners is likely the correct number for this sort of IA, perhaps a few more if the clinical coverage dictates it. There are too many partners, and management is stretched thin, which is reflected in the stage 2 evaluation.
- A company with prior experience with IVD or other medical devices is not only desirable but virtually required for regulatory work; you also need a device developer at its core.
- Clinical partners are not negotiable. Access to patient cohorts to the validation studies at least one or two hospitals or clinical research centers. This will be scrutinized by stage 2 reviewers.
- If you are addressing resource-constrained environments, an interviewee from a less developed nation or a multinational health agency has greater authority. It is not essential, but it makes the impact case much more impressive.
- Innovative SME should be included. A diagnostics SME or a startup with IP on sensor technology, sample prep components, or readout systems provides the consortium with a commercial footing not available to academic-only teams. (It is well to check twice: most of the IAs have been diluted by leaving commercial exploitation undefined.)
- The sightless assessment at stage 1 is peculiar and throws individuals off balance. Make sure that your Part B does not have any identifying information twice. Even indirect hints, such as referring to past EU-financed projects your team led, may constitute a technical breach of the rule.
- For stage 1, make the scientific narrative short and evaluator-friendly. The threshold is 4+4. The only criteria are Excellence and Impact. Do not interweave your argument on clinical differentiation in technical terms.
- In stage 2, the implementation criterion is added. It is important to have budget justification, work plan coherence, and risk mitigation. It is here that most proposals lose their points, not on science.
How would microfluidics contribute to this topic?
One of the weakest aspects of PoC diagnostics is the conventional sample preparation. It is usually manual, must be handled by a skilled person, and introduces variability that can lead to false results. Herein lies the issue that this call is meant to address. Microfluidics addresses it on the hardware side by integrating extraction, concentration, and detection into a miniaturized, reproducible workflow that the user does not need to think about.
You are at the bedside and want to distinguish between a bacterial and a viral infection, but do not want to order a sample sent to the lab. A microfluidic chip can perform multiplexed detection from a small amount of blood or saliva in less than 30 minutes. Different pathogen panel on the same platform. Such is what versatile means to this call.
- Miniaturization and integration: Microfluidic architectures intrinsically reduce the number of preparation steps to a single small cartridge, which falls within the dimension of equipment-free or equipment-modest, as in the REASSURED. Your consortium can mention this as a structural response to the Commission’s criteria, not as an attribute.
- Droplet microfluidics sensitivity: POC testing is known to be problematic for rare analytes in low-abundance samples. Droplet-based partitioning is highly sensitive to detection without limiting device size. In non-communicable disease monitoring, where biomarker concentrations may be very low, this is particularly important.
- Organ-on-chip platforms are also applicable, but not in the sense of a diagnostic device; rather, for validating the diagnostic device. It is possible to run cause-and-effect experiments on biological samples in a controlled microenvironment to verify what the device detects and what it does not. That is what the Commission needs as a comparative study.
- Compatibility with mass production: Microfluidic chips can be produced by polymer molding or by roll-to-roll processing at a reasonable cost. In a call that emphasizes resource-limited environments, the ability to demonstrate a plausible route to cost-effective large-scale production is truly beneficial.
The expertise that MIC has in the design of microfluidic chips, integration of sample handling, and development of PoC platforms directly translates to what this call is requesting. Not only does a technology provider or validation partner to the device architecture make you stronger, but having MIC at the design stage also strengthens the scientific case and the regulatory dossier.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ – HORIZON-HLTH-2027-02-IND-02-two-stage
What is HORIZON-HLTH-2027-02-IND-02-two-stage actually about?
The topic is the development of mobile, flexible point-of-care (PoC) diagnostic devices for use outside the lab. The Commission is not requesting additional research on the potential of PoC. It wishes for devices that are practical, meet EU regulations and can be used clinically. The objective is to move from laboratory to field conditions.
What kind of action is funded and how much budget per project?
What is the REASSURED framework, and why does it matter here?
The World Health Organization’s (WHO) REASSURED criteria describe what a PoC device should do: real-time connectivity, easy specimen collection, affordability, sensitivity, specificity, user-friendliness, rapidness, equipment-free or minimal, and deliverability to end-users. The Commission wants you to describe how you will meet each in the proposal. Using the REASSURED list as your proposal’s guide is the best way to demonstrate that you read (and understood) the call.
Why is blind evaluation used at stage 1, and what does it change for the writer?
Stage 1 evaluation is blind: no company names and/or acronyms, logos, or people names in the abstract or Part B. Why this matters: even the slightest suggestions, such as mentioning past EU projects your team has been involved in, might be considered a technical violation. Have a non-author clean the document twice before you submit.
Are US partners eligible? What about Chinese ones?
This topic is open to US entities due to NIH reciprocity. Entities that are controlled directly or indirectly by China are ineligible. This is a rare move in Cluster Health and it is best to be aware of this early in the process if you are considering a US entity. The eligibility line provides an opportunity to tap into US clinical or regulatory expertise.
What does the Commission mean by clinical needs assessment?
A clinical needs assessment here is more than a survey of clinicians. The Commission wants the whole care cycle and systems perspectives, not just the point-of-care perspective. Administrators, payers and end users (nurses or technicians) should be covered. This will be verified. We have seen the proposal’s score go down, which is a clinician questionnaire.
Should the device target infectious or non-communicable diseases?
Choose one or the other. If you want to target both infectious and non-communicable diseases in one device, it will be seen as “mission drift” or “creeping featurism”. For infectious diseases, viral, bacterial, and fungal causes are preferred. For non-communicable diseases, priority goes to life-threatening emergencies.
How early should regulatory compliance (IVDR/MDR) be addressed?
Day one. MDR or IVDR compliance is not a retrofit. Include some early thoughts on notified body contacts, intended use and risk classification in the project proposal. Projects that leave regulation to after the project rarely make it past stage 2.
What is a realistic consortium size for this Innovation Action?
8-12 partners, maybe a few more if you need to cover the clinical spectrum. More than that, and the project management becomes a drain on the implementation. A device developer, IVD or medical device company, at least one or two clinical partners, and an innovative SME with IP in sensors or readout systems. Including a partner from a low-resource country enhances the case’s impact.
Where does microfluidics make the strongest difference for this topic?
In the sample preparation. This is where traditional PoC fails. Microfluidics combines extraction, concentration and detection in a cartridge, with no manual processes and reproducible results. Droplet partitioning helps with low-concentration analytes. High-volume production of polymer chips makes the economics work. MIC’s chip design and sample processing know-how make a good idea a regulatory-compliant product.
