Tips & Tricks for a Successful HORIZON-HLTH-2026-02-DISEASE-12 Proposal
Opening
10 February 2026
Deadline
Keywords
ERDERA
Rare Diseases
disease pathogenesis
clinical decision-making
Pharmaceutical Strategy
Pharmaceutical Strategy
Organ-on-chip models
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
HORIZON-HLTH-2026-02-DISEASE-12: European Partnership on Rare Diseases (ERDERA) (Phase 2)
This is not a common open call. The second phase of ERDERA, the co-funded European Partnership on Rare Diseases, already exists and is operational, and has been granted funding through the Commission. Submission is limited to the current ERDERA coordinator. New partners are also welcome, and the transnational calls initiated by the partnership will finance dozens of third-party research projects. So, although you might not be a member of the current consortium, the subject concerns you.
Discover more!
Administrative facts: what do we know about the HORIZON-HLTH-2026-02-DISEASE-12 call?
Which call is it, and when is the opening and the deadline?
● Call name: Partnerships in Health
● Call identifier: HORIZON-HLTH-2026-02
● Destination: Tackling diseases and reducing disease burden
● Topic: HORIZON-HLTH-2026-02-DISEASE-12: European Partnership on Rare Diseases (ERDERA) (Phase 2)
● Opening date: 10 February 2026
● Deadline: 15 September 2026
● Type of action: Programme Co-fund Action (COFUND)
What about the budget and estimated size of the project?
● Overall budget: EUR 91.30 million (EUR 48.70 million from 2026 budget, EUR 42.60 million from 2027 budget)
● Number of projects expected: 1
● Budget for that single project: around EUR 91.30 million
● EU funding rate: 50% of eligible costs
● Financial support to third parties (FSTP): up to EUR 10.00 million per third party over the partnership duration
What are the key eligibility and evaluation conditions?
● Proposal must be submitted by the coordinator of the consortium funded under HORIZON-HLTH-2023-DISEASE-07-01 (“European Partnership on Rare Diseases”). Additional partners may be included.
● Award thresholds: 4 for Excellence, 4 for Impact, 4 for Implementation. Cumulative threshold: 12.
● JRC may participate as beneficiary with zero funding or as associated partner, but will not participate in proposal preparation.
● US entities are eligible for EU funding under NIH reciprocity. The consortium of research funders from eligible EU Member States and Associated Countries must expressly agree.
● Social sciences and humanities (SSH) contribution is required.
● Collaboration with JRC on rare disease registries is encouraged, especially around the EU RD Platform and pseudonymisation tools.
Deadlines of European Programmes (Version 12/2025)
Get the MIC Horizon Europe 2026/2027 Calls Calendar:
-All Horizon Europe deadlines (by cluster and call).

Scientific range: what the Commission expects from the HORIZON-HLTH-2026-02-DISEASE-12 grant
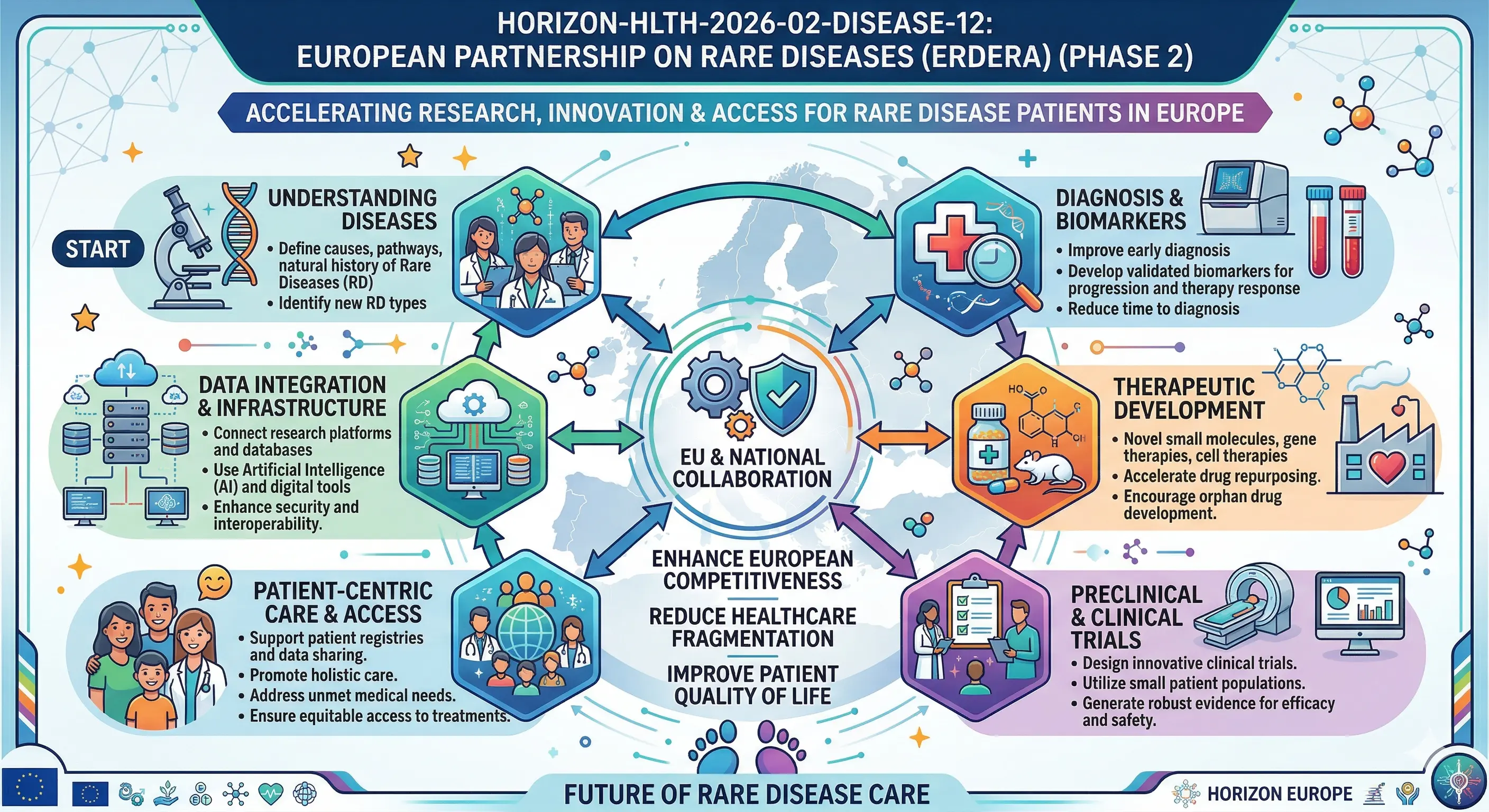
Continuity and scale-up is what the Commission is actually seeking. Phase 1 of ERDERA should build the structure. Phase 2 must broaden it, bridge the gaps, and provide patients with rare diseases with tangible results. Lists of publications will not do the trick. The evaluators desire to have operational impact on clinical research preparedness, information infrastructure, and coordinated inter-border funding.
Other activities (and other partners, should they be there) should be offered in the proposal as amendments to the current grant agreement. The collaboration will bring the following:
● Initiate rare disease R&I joint transnational calls, as part of the ERDERA Strategic Research and Innovation Agenda (SRIA), and fund patient-need-driven research with evident short, medium, and long-term contributions.
● Empower the European Clinical Research Network (CRN) to increase cross-border trial preparedness, based on European Reference Networks (ERNs), and demonstrate the role of the network in accelerating diagnostics and less costly treatment.
● Streamline the information ecosystem of rare diseases, through interoperable, federated, FAIR access to data, including registries, facilitating more efficient translational and clinical research. Applicable ESFRI research facilities need to be utilized.
● Combine the entire R&I spectrum of basic research to pre-clinical, clinical, and implementation research, eliminate overlaps, and set regional, national, and European priorities.
● Prevent, support, diagnose, and treat with coordinated training, implementation research, and stakeholder engagement, and encourage adoption of health innovations in clinical practice.
● Collaborate with the International Rare Disease Research Consortium (IRDiRC) to strengthen the European contribution to the world and prevent duplication.
The collaboration is also supposed to support the goals of the Pharmaceutical Strategy for Europe and the European Health Data Space. Simply put, it concerns linking research, regulation, data and patients within a single logical system.
Scientific strategy: How can you enhance your chances of being funded through HORIZON-HLTH-2026-02-DISEASE-12?
● Demonstrate significant improvement since Phase 1. Don’t just list outputs. Show what happened to the patients, to be trial ready, to have access to the data.
● Not plans alone, but evidence of multi-national trial coordination, must be provided by the CRN element. Evaluators will seek to find evidence that the network is in fact happening.
● FAIR data integration is one of the priorities which the Commission continues to reiterate. When the ecosystem of data is still in bits, it is a warning sign.
● Engaging the patient as co-creators, not as window dressing.
● Hold the SRIA in mind. All new actions are to be mapped to it.
Consortium & proposal-writing plan: what works best with this type of Health RIA?
● The consortium has already been established. The question is whether to invite other partners and who.
● When your organisation holds a rare disease registry expertise, clinical trial infrastructure or ERN membership, there is a real opportunity available to you as a new partner.
● A new SME that provides a diagnostic or data interoperability tool would make a difference here and specifically the FSTP-funded transnational calls.
● The proposal technically is an amendment of a grant agreement. The format of writing is not the same as a typical proposal. Look at what is new, what is extra and why it cannot be accomplished by the current consortium on its own.
● The use of SSH is not optional. Budget it properly.
How would microfluidics contribute to this topic?
An inherent bottleneck to rare disease research is that the number of patients is minuscule and biological samples are limited and traditional assays consume material rapidly. Microfluidic platforms alter that equation. Candidates of drugs can be screened on microlitre of patient cells. You run ten conditions where you used to run two
● Organ-on-chip models allow you to recapitulate rare disease phenotypes on a chip when animal models do not exist or do not scale to humans. Suppose you are dealing with a rare metabolic disorder where no mouse model has been validated. A liver-on-chip cultured with patient iPSC-derived hepatocytes provides you with a testbed of the disease.
● Minimal blood volumes can be processed on diagnostic microfluidic cartridges and this is crucial in paediatric rare diseases where every draw counts.
● Microfluidic screening of orphan drug candidates under high-throughput conditions is accelerating the therapeutic pipeline, and that directly fulfills the requirements of the CRN under ERDERA.
● In registry-linked biobanks, microfluidic sample preparation minimizes pre-analytical variation, and as a result, your biomarker data is comparable across sites and around the world.
Should your consortium want to apply to ERDERA transnational calls, it boasts a microfluidics partner that embodies the type of tool innovation the Commission would like to see become a part of clinical practice. That’s not a nice-to-have. In rare diseases, where each sample counts and each patient counts, it can be the difference between a successful proof of concept languishing in a laboratory notebook and one that makes it to a clinical workflow.
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
