Tips & Tricks for a successful HORIZON-HLTH-2026-01-DISEASE-11 proposal
Opening
10 February 2026
Deadline
Keywords
Cluster health
risk factors
RIA
disease Prevention
cardiovascular diseases
gender-specific determinants
targeted interventions
hormone-linked mechanisms
Your microfluidic SME partner for Horizon Europe
We take care of microfluidic engineering, work on valorization and optimize the proposal with you
HORIZON-HLTH-2026-01-DISEASE-11: Understanding of sex and/or gender-specific mechanisms of cardiovascular diseases: determinants, risk factors and pathways
In Europe, cardiovascular diseases are the leading cause of death and 1.71 million EU citizens died of circulatory causes in 2021, approximately 32% of all deaths. What the Commission is after here is one thing: mechanistic explanations of the way sex and gender so disparagingly contribute to CVD. Then what do we do with that knowledge. This RIA finances that work, biologically all the way to tools that clinicians can literally put to practical use.
Download the MIC Horizon Europe 2026/2027 Calls Calendar:
Discover more!
Administrative facts: what do we know about the DISEASE-11 call?
Which call is it, and when are the opening and the deadline?
- Call name: Cluster 1 – Health (Single stage – 2026)
- Call identifier: HORIZON-HLTH-2026-01
- Destination: Tackling diseases and reducing disease burden
- Topic: HORIZON-HLTH-2026-01-DISEASE-11 – Understanding of sex and/or gender-specific mechanisms of cardiovascular diseases: determinants, risk factors and pathways
- Opening date: 10 February 2026
- Deadline: 16 April 2026, 17:00 Brussels time
- Type of action: Research and Innovation Action (RIA)
What about the budget and estimated size of the project?
- Overall topic budget: EUR 39.30 million
- Indicative number of funded projects: 6
- Expected EU contribution per project: EUR 6.00 to 7.00 million
What are the key eligibility and evaluation conditions?
- Evaluation thresholds: Excellence 4, Impact 4, Implementation 4. Cumulative threshold: 12.
- Legal entities established in China are not eligible to participate in RIAs and IAs under this destination.
- In recognition of the opening of the US National Institutes of Health programmes to European researchers, US entities are eligible for EU funding in most 2026 topics.
- The JRC is not explicitly mentioned as a potential partner in the DISEASE-11 topic text (unlike, for instance, TOOL-03 or STAYHLTH-03).
- No mandatory clustering requirement has been identified for DISEASE-11. Clustering applies to ENVHLTH-01, ENVHLTH-04, and TOOL-03 but not here.
- The topic requires effective contribution of SSH disciplines and experts.
- FAIR data principles and GDPR-compliant data sharing are expected.
- Clinical studies, if included, require the dedicated annex.
Scientific range: what does the Commission expect from the DISEASE-11 grant?
The differences between sex and gender influence the appearance of CVD, the response of patients to treatment, and its outcome. Pregnancy complications, menopause, risks with hormones: it is a female specificity that is poorly treated even unnoticed. The majority of the care paths do not consider any of that. The Commission would like that to be fixed.
What are the results requested by the work programme?
Improved awareness of sex and gender-specificity in determinants and pathways of CVDs. Not only to be published, to make it available to clinicians and those who manage interventions.
Model risk validated sex and gender that enhances our detection, prevention and treatment of CVD. The term validated is carrying its weight here. Retrospective subgroup plots will not do.
Plans that in fact decrease the CVD strain with sex and gender incorporated into the design, rather than as an interim thought.
What are the areas of scientific directions?
The majority of these should be covered in proposals:
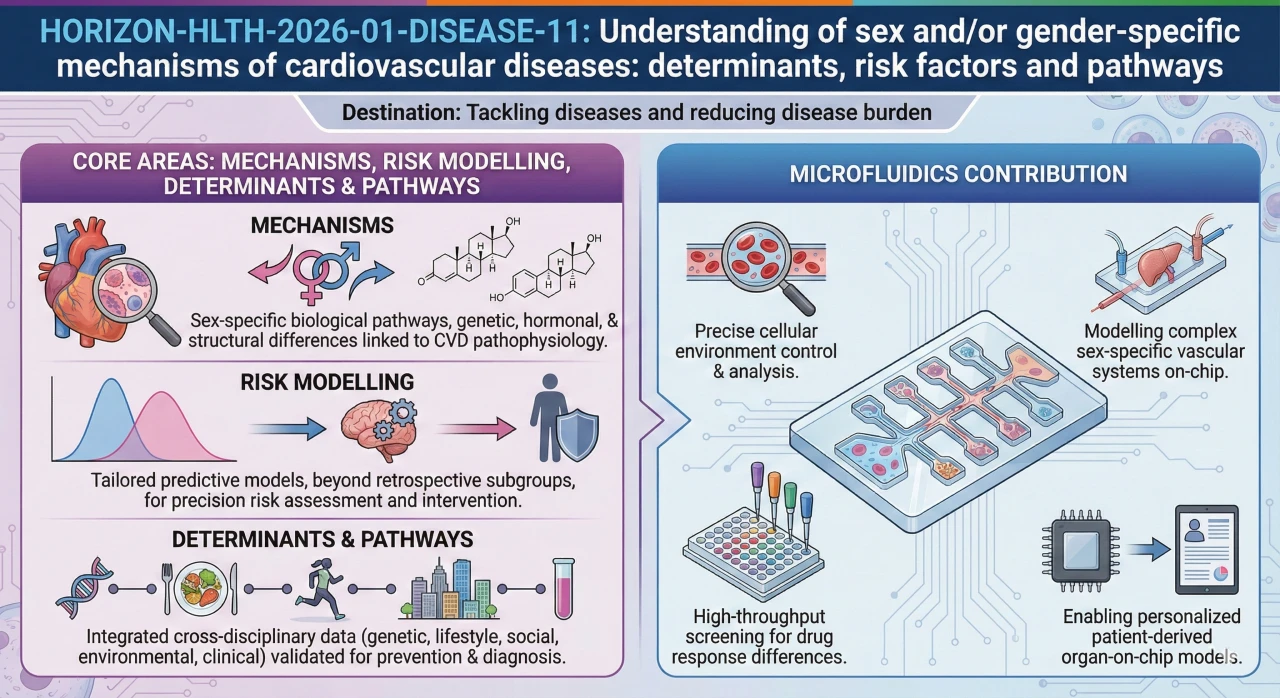
- Mechanisms: What are the hormonal, structural, and biological sex differences that are connected to the CVD pathophysiology? The work on endothelials, inflammation, and blood coagulation answers such questions.
- Risk modelling: Construct and confirm sex-specific models.
- Determinants and pathways: Affirm sex- and gender-specific variables through molecular biology, behavioural science, clinical information, epidemiology, and genetics.
It must not have AIs where they do not solve a real problem (not yet a buzzword). Intersectional approaches are overtly solicited. And propositions should demonstrate a clear connection between the science, and something that can be of use to a clinician or a public health responsibility.
The thing is, the Commission does not want a collection of papers. They desire evidence that is practice-changing.
Scientific strategy: how can you enhance your chances of being funded through DISEASE-11?
Which scientific decisions are the most important?
- Not only variables, but also mechanistic drivers here are sex and gender. The biology part is sex, the rest of it is all layers imposed on it by the society. All of them require their respective hypothesis and their dataset. We have observed that proposals have performed poorly since they have considered gender as a filter in the section of statistics.
- Create a causal path through which evaluators can track. Pathways are caused by determinants, biomarkers are caused by pathways, risk models are caused by biomarkers, prevention decisions are caused by risk models. In case of missing any link or hand-wavy, the Excellence panel notices.
- The risks that are unique to women should be given a dedicated work package. Complications during pregnancy, influence of menopause, hormone influence. These do not need to be buried under an aside goal. Give them space.
- Turn your risk models into realists. External justice, calibration, fairness. And put it down in concrete terms regarding what is altered in practice as a clinician uses the model (check twice before you write).
- SSH is not a checkbox to design: SSH is a real partner. When SSS is the sole result of the dissemination WP, then that will normally cost you points.
- On AI: What bottleneck does it solve? Bias, what data, what problem, how do you deal with it. It is likely that three sentences will be sufficient, but they must be particular.
Consortium & proposal-writing plan: what works best with this type of Health RIA?
- Aim for 10-15 partnerships. Mechanisms, epidemiology, clinical work, and translation EUR 6-7 million. Not many enough partners and you shall have blind spots. Reviewers and too many begin to ask questions of your management budget.
- Three pillars that you must have in place: cardiovascular clinicians who have access to cohorts of patients, epidemiologists with access to registries, and molecular biologists who perform omics and hormone. Miss any of them and evaluators will mark.
- It is not necessary to leave the lab and turn to patients at once. Risk modelling requires you to have a data science group. SSH scholars that influence the study design, and not merely comment on it. Patient groups: Trials of whether strategies are workable. We have visited this excursion at least as many consortiums.
- At least one innovative SME should be included, in case possible. Making clinical software, decision support, analytics. Enriches your exploitation department and the work programme stimulates it.
- Reflect the call in your WP structure. WP on determinants/pathways, WP on risk model building, WP on translation, WP on SSH and stakeholders. It is a merit to the evaluators that the proposal sounds as a response to the question they posed.
- Construct a question driven story. What mechanism? What changes for patients? How does adoption work? And provide a logic model (one page). The majority of human beings overlook it, though in our case it is useful than you would imagine.
How would microfluidics contribute to this topic?
Cohort studies inform you on what occurs at population level. They don’t tell you why. You require a controlled setup in the event that your consortium should demonstrate that oestrogen safeguards the vessel wall, or that male and female immune cells react in different ways. That is where organ-on-chip comes in.
- Suppose your clinical partners have cohort data regarding premenopausal women with lower rates of cardiac events. Why? Put serum on a vascular chip, and run it on and off oestrogen, and you have a mechanistic answer such as could never be given by the cohort alone. A single experiment, yet it can support a whole WP.
- One of the major questions in this call is the instability of plaque. The immune element acts in a different way by sex although it is almost impossible to separate it out in a patient study. On a vascular chip in control of immune cells you can do it all.
- Another angle is considered the drug response. Cardiac tissue chips also enable you to compare the response of male and female tissue to the identical compound. Same drug, different outcome. When evidence becomes so concrete, it is typically the attention of excellence reviewers.
- At the omics level: molecular analysis of pair chip experiments. Cleaner signatures are due to controlled conditions. Of much importance when feeding such into a risk model later.
- And here is one of the points that should not be overlooked. Risk model inputs can take the form of the data of chip experiments as interpretable features (this connects your bench work to population level prediction and typically also enhances Excellence and Impact).
The MIC already brings its expertise in microfluidics to Horizon Europe:
H2020-NMBP-TR-IND-2020

Microfluidic platform to study the interaction of cancer cells with lymphatic tissue
H2020-LC-GD-2020-3

Toxicology assessment of pharmaceutical products on a placenta-on-chip model
FAQ - HORIZON-HLTH-2026-01-DISEASE-11
What is HORIZON-HLTH-2026-01-DISEASE-11?
It is a Research and Innovation Action (RIA) within the Health Cluster of Horizon Europe that will produce actionable evidence on sex- and/or gender-specific determinants, risk factors, and pathways in the cardiovascular diseases (CVDs) and translate the acquired knowledge into more effective prevention, detection, diagnosis, and treatment strategies.
What are the most important administrative particulars?
Opening date: 10 February 2026
Deadline: 16 April 2026
Total budget: EUR 39.30 million
Expected number of projects: 6
Budget per project: EUR 6 00-7 00 million.
Type of Action Research and Innovation Actions (RIA).
What is the significance of this issue to the EU?
In Europe, cardiovascular diseases are still a major killer of people, and in 2021, 1.71 million people died in the EU (32% of all deaths). Sex and gender disparities have important impacts on the symptoms of CVD, the prevalence, the response to treatment, and its outcomes. Certain conditions such as menopause or pregnancy complications are unique to women, thus women-specific interventions might be more effective but fail to take these vital prevention and care avenues into consideration.
What is expected in the way of scientific work?
Projects are supposed to cover a large portion of the following in realistically:
-Mechanisms: Determine the structural, hormonal, and biological differences between sexes/genders with CVD pathophysiology.
-Risk modeling: Test and establish sex or/and gender risk models.
-Determinants and pathways: Discover and confirm determinants that are sex- and/or gender-specific based on combined, multidisciplinary data (molecular biology, behavioral science, nutrition, clinical research, epidemiology, genetics/epigenetics).
What can the microfluidics do to this subject?
Microfluidics can be used as a proof engine of sex- and hormone-linked mechanisms:
-Vessel-on-a-chip/endothelium-on-chip models: Sex-hormone modulators of endothelial dysfunction, inflammation, and thrombosis.
-Immune-vascular microphysiological systems: Sex-differentiated immune signaling towards plaque instability.
-Microfluidic platforms of cardiac tissue: Experiment sex-specific drug response and electrophysiological variation.
-Microfluidic integration with omics: Create controlled multi-omics signatures in biomarker discovery.
-To risk models: Predictive models Use chip-derived mechanistic parameters as interpretable features.
What do you consider the pitfalls to avoid?
-The search to attempt to treat sex/gender as sub group analysis instead of as mechanistic drivers.
-Hyping clinical effects with no implementation routes.
-The experts in the field of SSH perceive the expertise as an aid in dissemination instead of part of study design.
-Absence of external validation, calibration and checks on fairness of the risk models.
-Failure to discuss the nature of the incorporation of risk models into clinical guidelines or practice.
What is the type of consortium that is likely to score well in DISEASE-11?
The suggestions that are comfortable to appraisers tend to have a triangular form with a bridge:
- Triangle (core science):
Cardiology clinical skills (hospital networks, clinical cohorts).
Epidemiology/public health (registries, population cohorts, exposure science).
Molecular/systems biology (omics, genetics/epigenetics, hormone biology).
- Bridge (translation/adoption):
Artificial intelligence/data science/biostatistics (risk model development, validation, design of deployment).
SSH knowledge (gender studies, behavioural science, inequalities) influencing study design- not only dissemination.
Patient organisations + prevention stakeholders (uptake, relevance, ethics, feasibility).
A discreet but often effective move: include an innovative SME that can turn evidence into something deployable (validated pipelines, decision-support prototypes, clinical-grade tooling, or lab-to-model integration). It tends to strengthen the “Impact and implementation” spine of the story. The Microfluidics Innovation Center is an SME specialized in microfluidic engineering and valorization of proposals in Horizon Europe.
